Advising suckler herds on IBR control following Beef Welfare Scheme testing
Maria Guelbenzu LV PhD MRCVS, BVD programme manager, AHI, provides vets with some guidance in relation to interpreting the National Beef Welfare Scheme Infectious Bovine Rhinotracheitis test results and advising their clients about them
The National Beef Welfare Scheme (NBWS) is a support measure introduced by the Department of Agriculture, Food and the Marine (DAFM) designed to enhance animal health and husbandry on Irish suckler farms in 2023. The scheme supports farmers in meal-feeding suckler calves in advance of, and after, weaning, and in testing for the presence of Infectious Bovine Rhinotracheitis (IBR) in their herds. Both actions are mandatory for farmers who wish to participate in the scheme. Results from this testing are currently being analysed and will be reported in due course.
TESTING A PROPORTION OF THE HERD FOR IBR: THE SNAPSHOT
The IBR testing of a proportion of the herd (snapshot test) in the NBWS 2023 is a cost-effective means to obtain an initial indication of the prevalence of infection and the effectiveness of any control measures currently in place in a given herd. The snapshot requires the sampling of 20 randomly selected animals — used or intended for breeding and over nine months-old — to ensure there are no maternal antibodies remaining. It is important to include animals of all ages and groups in this testing to obtain a result that truly reflects the status of the herd. Blood samples are tested with an IBR gE (marker) test that allows differentiation between vaccinated and infected animals.
Having an indication of the within-herd prevalence to IBR, together with analysis of the age of seropositive animals where detected, provides useful information to review the biosecurity of the herd to prevent re-introduction of the disease and make informed decisions on vaccination.
Relationship between the snapshot results and within-herd prevalence
A direct correlation exists between the proportion of positive animals detected in the snapshot test and within-herd prevalence. To explore this, the relationship between the snapshot test results when sampling 20 randomly selected animals and the apparent within-herd prevalence was assessed with the ‘Grange beef dataset’ which consists of 131 herds sampled in 2015, where all adult animals were tested for field virus antibodies. It found 86 per cent herd level prevalence (113 positive herds). Of the positive herds, 37 herds had more than 30 animals. For each of the 37 herds with more than 30 animals and known apparent prevalence within the herd, the snapshot was simulated 10,000 times.
Using these snapshot test results, the within-herd prevalence was estimated and compared to the known prevalence within the herd. This comparison is shown in Figure 1. For each herd, the entire range of estimated prevalences from the snapshot is shown as a vertical red line and compared to the measured prevalence within the herd calculated from the full herd test results (red squares). The mean deviation for the 20-animal scenario is approximately five per cent, which means that a snapshot overestimated or underestimated the true prevalence within the herd by five per cent on average, with the greatest variability seen with around 50 per cent prevalence.
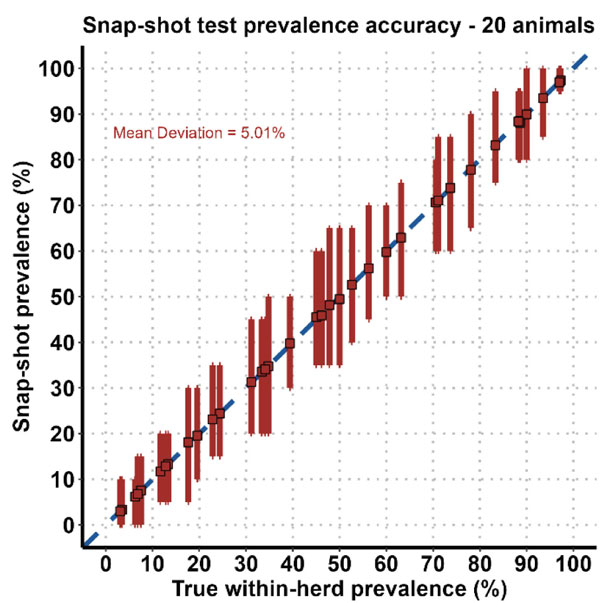
Figure 1: Snapshot test prevalence accuracy based on 10,000 random samplings for each herd.
How long does it take for a herd that had an outbreak to get to a low within-herd prevalence of 10 per cent?
As an initial assessment, the rate of animal turnover in beef herds was simulated from national data, as shown in Figure 2, showing the proportion of these animals remaining in each subsequent year. This analysis shows that, if considering a herd with an initial seroprevalence of 100 per cent, after a median time of approximately nine years, only 10 per cent of the original herd remains (with a prevalence of 10 per cent assuming no further virus circulation or introduction of positive animals).
This is further illustrated in Figure 3, noting that nine years is a median value (the number of years for 50 per cent of herds to achieve the 10 per cent value). This figure shows the proportion of herds reaching the 10 per cent value in year nine (around 15 per cent of herds), or in fewer or more years, and in addition, the number of original animals remaining at that point (median values of five or fewer).
This demonstrates that it takes many years for a herd that had an outbreak to achieve a low prevalence or fully negative status. Therefore, we can assume low prevalence/negative herds have been free from IBR for a long period of time, and they have avoided accidental introduction of IBR or virus positive animals.
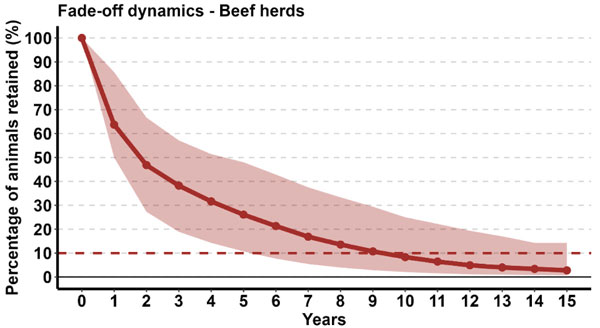
Figure 2: Population turnover in suckler beef herds. The line represents the median proportion of animals still in the herd in each successive year (e.g., 10 per cent remaining after nine years) while the wider band indicates the upper and lower limits within which 95 per cent of values lie (e.g., reading vertically, after 10 years, 95 per cent of herds have fewer than 25 per cent of animals remaining; reading horizontally at 10 years, 95 per cent of herds require at least five years before only 10 per cent of the starting population remain).
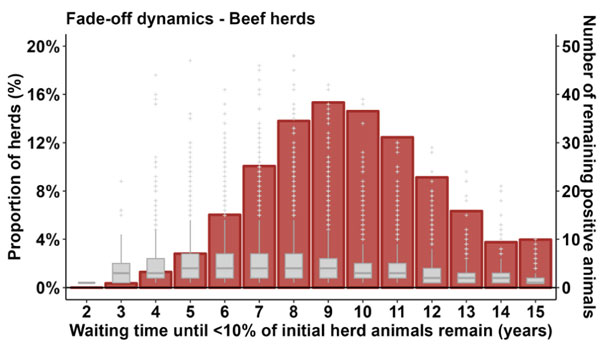
Figure 3: Fade off dynamics of seroprevalence to IBR in beef herds starting from an initial 100 per cent showing the number of years until less than 10 per cent of the initial herd remains. Vertical red bars represent the proportion of herds (left y axis) in which less than 10 per cent of the starting population remain after a given number of years, while the grey box plots indicate the distribution of the number of animals remaining when the 10 per cent threshold is reached (right y axis) and could be removed to achieve freedom.
Porcine Dermatitis Nephropathy Syndrome (PDNS)
PDNS cases have irregular red/purple macules and papules on the skin and will be anorexic and depressed (Figure 3). They normally do not have a fever. They may be reluctant to move, and/or be stiff-gaited. PDNS may be diagnosed if the following criteria are met:
Presence of haemorrhagic and necrotising skin lesions, and/or swollen and pale kidneys with generalised cortical petechiae;
Presence of systemic necrotising vasculitis and necrotising, fibrinous glomerulonephritis;
Detection of PCV2 is not indicated as diagnostic for PDNS.
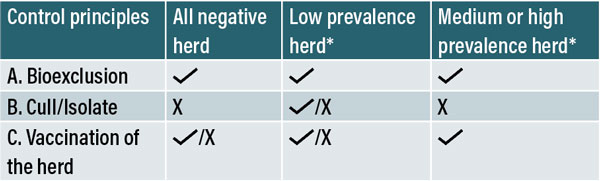
Figure 4: Methods for IBR control a per within-herd prevalence.
ADVICE FOR HERDS WITH MEDIUM-HIGH PREVALENCE > 15 PER CENT
Non-vaccinating herds
Herds with a snapshot indicating a within-herd prevalence of >15 per cent that are not already vaccinating for IBR should consider beginning a vaccination programme. An effective vaccination programme will reduce the likelihood of latent infection in carrier animals reactivating and resulting in transmission within (and potentially between) herds. It will also protect animals that become exposed, reducing the extent and duration of any clinical signs and reduce the level of onward transmission from these animals. Over time therefore, as latently infected cows are culled through routine management, and replaced by young, unexposed animals, the prevalence of infection in the herd will fall, potentially reaching zero depending on various factors, including the level of biosecurity and purchase policy.
Vaccinating herds
Herds with a snapshot indicating a within-herd prevalence of >15 per cent that are already vaccinating for IBR, should consider evaluating the efficacy of their overall control strategy (vaccination and biosecurity), taking into account the estimated prevalence and number of years vaccinating.
This should include quarantine and testing of added animals to ensure they are not carriers, and strategic sampling of younger stock that should not have been exposed if vaccination and associated biosecurity is 100 per cent effective. For example, in a herd that has been vaccinating for five years, we would like to see all home-bred animals less than five years of age testing negative.
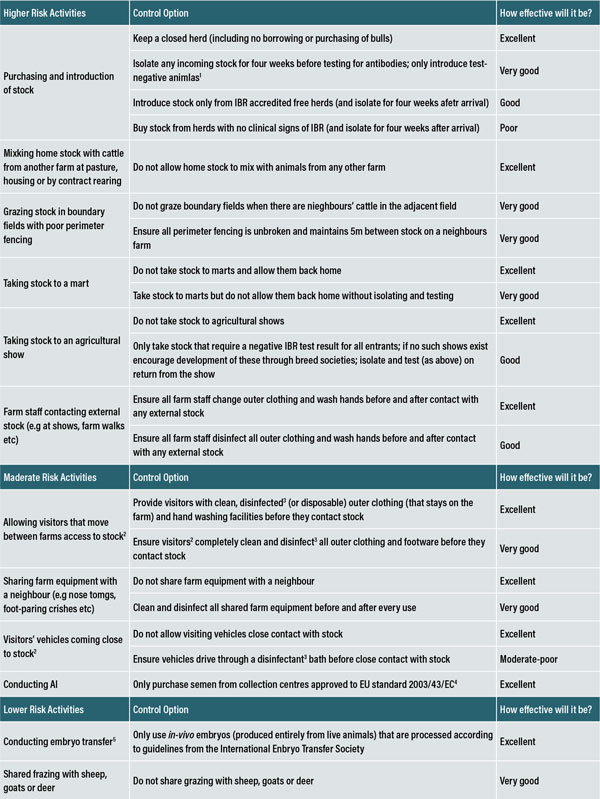
1If introducing stock follow the Bioexclusion document guidelines, and see www.animalhealthireland.ie for more information on testing animals. 2This includes delivery/pick-up and their helpers. 3A list of approved disinfectants is available for the websites of DAFM (www.agricuture.gov.ie) and DARD (www.dardni.gov.uk/dard-approved-disinfectants). 4All semen collection centres in the Republic of Ireland and all legally imported semen must meet these standards. 5This does not include the risk of purchasing recipients, which is a ‘higher risk’ activity (considered above).
Figure 5: Bioexclusion control options for activities that present high, moderate and low risks of introducing IBR into a herd.
ADVICE FOR HERDS WITH LOW/NO PREVALENCE (0-15 PER CENT)
Herds with a totally negative snapshot or with a single seropositive animal have a very low (or zero) true within-herd prevalence. Testing of any animals not included in the snapshot would be required to confirm freedom (or to identify and remove any remaining carriers). Again, those in charge of these herds should review biosecurity, particularly in relation to added animals, to avoid accidental introduction. Vaccination should be considered but not necessarily introduced if the risk of introduction can be minimised with bioexclusion measures, noting that at this point the herd has avoided introduction of infection for many years.
It is worth highlighting that, under the Animal Health Law, for a herd to be considered IBR-free as well as testing negative, it must not have been vaccinating for at least two years.
INDIVIDUAL ANIMAL RESULTS
Analysis of the results at individual animal level will provide an indication of whether there has been recent circulation of the virus and, therefore, if control measures are effective in herds with an IBR control plan. The first step is to look at the ages of any seropositive animals and their cohorts. If only older animals are positive, it indicates historical infection and good control, as the virus has not circulated to younger animals. If only young animals are positive, it indicates recent infection.
It is also important to note whether any seropositive animals are home-bred or not. If they are not home-bred, consider whether they could have received a conventional vaccine. Although only ‘marker’ vaccines are licensed for use in the Republic of Ireland, in other countries (including the United Kingdom) both ‘conventional’ (non-marker) and ‘marker’ vaccines are available. Animals vaccinated with a conventional vaccine will test positive to the gE (marker) test.
BIOSECURITY REVIEW
A review of the risk activities and possible control options can be seen in Figure 5. Closed herds have the lowest risk of introducing IBR but if the farmer has to introduce animals (including animals returning from shows or the mart), these should be isolated for four weeks and tested for IBR antibodies before they join the herd. Only negative animals should enter negative herds.
Avoiding mixing ‘home’ stock with cattle from other farms at pasture, housing or during contract-rearing, will help prevent accidental introduction of infection. As the virus is also capable of being transmitted indirectly through equipment and people, it is important to maintain good hygiene of shared equipment and facilities, use separate clothing, or ensure appropriate cleaning and disinfection of boots and clothing.
Good building design, ventilation, and stocking density, as well as ensuring good nutrition and low stress environments, are key.
Further information
Detailed information leaflets on IBR and herd biosecurity, along with answers to frequently asked questions on IBR and specific guidance for herds with bull calves that are potential AI sires, are available at
http://animalhealthireland.ie/?page_id=377.
1. What constitutes an IBR snapshot test:
A. Consists in sampling a fraction of the herd
B. Within the NBWS, 20 animals are blood sampled
C. Animals should be randomly selected and over nine months old
D. All of the above
2. What statement is not true in relation to the IBR snapshot test?
A. In order for the results to be representative, sampled animals must be randomly selected
B. A direct correlation exists between the prevalence detected at the snapshot test and within-herd prevalence
C. Oldest animals should not be sampled
D. When sampling 20 animals, a snapshot overestimated or underestimated the true prevalence within the herd by five per cent on average
3. A herd with a negative snapshot
A. Is likely free, but if pursuing freedom, all animals would have to be individually sampled
B. Has not had introduction of IBR for a number of years
C. Vaccination should only be considered if higher risk activities for IBR introduction are not controlled
D. All of the above
4. What would you advise a herd that has positive gE snapshot test result?
A. Concentrate on bio-exclusion
B. The herd is free from infection
C. Start full and regular vaccination combined with bioexclusion and regular monitoring
D. Start vaccination of the breeding animals in the herd
ANSWERS: 1D; 2C; 3D; 4C.
















