Canine degenerative mitral valve disease in dogs
Julie Marner Bilchak MVB MRCVS, Commercial Technical Manager, Norbrook Laboratories, provides an overview of the significant changes that have taken place during the last decade in the staging, monitoring, and treatment of degenerative mitral valve disease in dogs
Canine heart disease is frequently encountered in veterinary practice, the most common form of which is chronic degeneration of the leaflets of the mitral valve. Historically, management of degenerative mitral valve disease focused on improving quality of life for patients once they had entered congestive heart failure, with no good treatment or monitoring recommendations for subclinical disease. As a result, discovering a heart murmur in an otherwise healthy older patient was frustrating for the veterinarian and the owner. Over the past ten years research has resulted in significant changes to how we stage, monitor, and treat patients with this condition, especially those patients presenting with subclinical heart murmurs.
Canine mitral valve disease pathogenesis and epidemiology
Chronic myxomatous degeneration of the cardiac valves is an acquired form of heart disease most commonly affecting the leaflets of the left atrioventricular (mitral) valve. This disease is referred to interchangeably as degenerative mitral valve disease (DMVD) and myxomatous mitral valve disease (MMVD). The right atrioventricular (tricuspid) valve is also involved in an estimated 30 per cent of cases1.
This disease is characterised by thickened plaques and nodular changes to the leaflets of the valves and chordae tendinae, contraction of the leaflets, and rolling of the free border1. The lesions can render the leaflets unable to make good contact with each other and, therefore, unable to maintain a tight seal, resulting in mitral regurgitation.
Although prevalence estimates differ, DMVD is accepted as being the most common form of acquired heart disease in dogs. A 2015 study scrutinising electronic medical records from 111,967 dogs attending 93 primary-care practices in England reported a 3.9 per cent prevalence of confirmed and suspected cases of DMVD2.The mean age at which disease was first detected in this study was 9.5 years and the mean body weight was 10.9kg, confirming the accepted epidemiology that this is a disease of middle-aged to elderly dogs and more prevalent in dogs weighing less than 20kg. Males were more likely than females to be diagnosed in this study, which is again consistent with previous studies3. A strong breed predisposition has been recognised for DMVD, and in this study population, breeds which had a statistically significant increased risk included Cavalier King Charles Spaniels, King Charles Spaniels, Chihuahuas, Whippets, Poodles, Shih Tzus, Miniature Schnauzers, and Border Collies.

Table 1. Six-point grading scale for canine heart murmurs.
Describing and grading heart murmurs
Auscultation of the healthy canine heart should reveal two clear and distinct heart sounds. The first heart sound (S1) corresponds to the closure of the atrioventricular (mitral and tricuspid) valves at the beginning of systole, and the second heart sound (S2) corresponds to the closure of the semilunar (pulmonic and aortic) valves at the beginning of diastole. Normal heart anatomy and blood viscosity results in laminar flow of blood between the heart chambers and through vessels (which is not audible). However, cardiac pathology or changes to blood viscosity may cause turbulence in blood flow resulting in vibrations of anatomical structures which can be detected as an audible murmur, or when more pronounced, a palpable “thrill”. When a murmur is detected, the location of maximal intensity and timing in the cardiac cycle, combined with signalment, can help give a strong suspicion of the underlying pathology which is causing the murmur.
Timing in the cardiac cycle – Mitral insufficiency will result in a systolic murmur, heard between S1 and S2 heart sounds. Mild mitral regurgitation may be associated with an early systolic murmur whereas a holosystolic murmur is more likely to indicate advanced pathology4.
Murmur location – A murmur caused by mitral insufficiency will have its point of maximal intensity on the left side of the thorax and towards the apex of the heart. Timing and location can help localise the murmur to the mitral valve, and a list of differential diagnoses can be made including DMVD, mitral valve dysplasia, endocarditis, and dilated cardiomyopathy. Echocardiography is required for definitive diagnosis, but an understanding of epidemiology can help narrow differentials. For example, a young, large breed patient with a systolic left apical murmur is more likely to have congenital mitral valve dysplasia than DMVD. Alternately, when a large breed dog develops an acquired heart murmur attributed to the mitral valve, the differential diagnoses should include dilated cardiomyopathy and endocarditis as well as DMVD5.
Murmur grade – Although alternative murmur grading systems have been proposed, most veterinarians are familiar with a grading system based on a scale of 1-6 using features such as the relative intensity of the murmur sound when compared to the normal heart sounds, and the presence or absence of a palpable thrill6. In the case of DMVD, the intensity or “grade” of the murmur provides additional information because it correlates with the severity of the mitral regurgitation7. A murmur greater than or equal to Grade 3 comes with a higher risk of being haemodynamically significant and resulting in cardiomegaly. It is important to note that this correlation between murmur grade and severity of pathology is not seen with other acquired cardiac conditions like dilated cardiomyopathy, nor is it true of congenital heart defects.
ACVIM staging systems for canine mitral valve disease
The American College of Veterinary Internal Medicine (ACVIM) Specialty of Cardiology consensus panel adopted a staging system for canine DMVD in 2009 and further refined it in 20198,9. This staging system replaces previous systems like the New York Heart Association (NYHA) Functional Classification, which is still widely used in human cardiology, but has not proven to be a good framework around which to build veterinary treatment recommendations. The ACVIM staging system defines patients as having preclinical “heart disease” when cardiac pathology is detected, usually in the form of an audible murmur. The term “heart failure” is reserved for patients with cardiac clinical signs. Patients can be grouped into stage A, B, C or D and it is expected that an animal will progress from one stage to the next over time unless corrective cardiac surgery is undertaken. This staging system forms the framework around which the ACVIM has built practical diagnostic and treatment recommendations.
Stage A diagnosis
Dogs are in Stage A DMVD if they are at high risk of developing DMVD based on breed predisposition, but do not have any known cardiac pathology at the time of evaluation. For example, a Cavalier King Charles Spaniel with no audible heart murmur.
Stage A treatment recommendations
There are currently no known interventions that will prevent these animals from developing cardiac pathology later in life.
Stage B diagnosis
Dogs are in Stage B DMVD when they have evidence of mitral valve disease (typically recognised as an audible heart murmur) but do not have any cardiac clinical signs. Classically, these heart murmurs are picked up incidentally during an annual wellness visit or pre-anaesthetic exam. Some animals with a DMVD heart murmur will never develop clinical signs of congestive heart failure because their mitral degeneration is occurring at such a slow rate that they are likely to die from another cause before heart failure occurs. For others, the mitral regurgitation will worsen over a shorter period, becoming haemodynamically significant and causing remodelling of the heart chambers before progressing into congestive heart failure.
The ACVIM staging system divides Stage B into two subgroups: stage B1 which are patients with subclinical disease and no significant evidence of cardiomegaly, and Stage B2 which are subclinical patients with a heart murmur greater than or equal to Grade 3 and evidence of significant cardiomegaly.
Figure 1: Lateral thoracic radiograph without annotations. Radiograph courtesy of Nigel Johnson MVB.
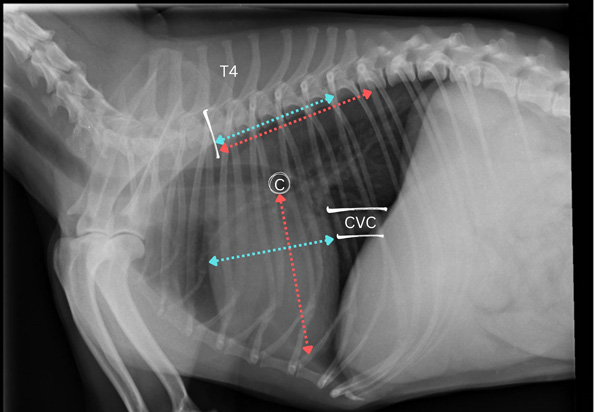
Figure 2: VHS. Lateral thoracic radiograph with carina (c), caudal vena cava (CVC), and spinous process of the fourth thoracic vertebra (T4) identified. Long axis of the cardiac silhouette (red line) is measured from carina to the apex of the heart and short axis (blue line) is measured from the cranial to the caudal border. These measurements are expressed in terms of vertebral body units using the cranial border of the fourth thoracic vertebra as the starting point.
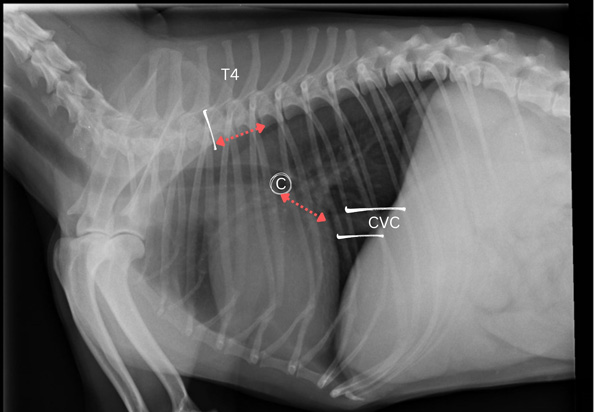
Figure 3: VLAS. Lateral thoracic radiograph with carina (c), caudal vena cava (CVC), and spinous process of the fourth thoracic vertebra (T4) identified. Distance is measured from the carina to the dorsal border of the caudal vena cava at the site where it intersects the caudal aspect of the cardiac silhouette (red line). This measurement is expressed in terms of vertebral body units using the cranial border of the fourth thoracic vertebra as the starting point.
It is important, therefore, when Stage B patients are identified to take steps to discern whether significant cardiac remodelling is occurring, as this will have implications for how we discuss prognosis with owners, and for treatment recommendations. Echocardiography is the most sensitive means of determining whether chamber sizes are within normal limits, but thoracic radiography can also be used. When echocardiography is available, patients should meet all three of the criteria below for their cardiac remodelling to be considered significant enough to place them in Stage B2:
- Heart murmur ≥ Grade 3;
- Echocardiographic left atrium: aortic (LA: Ao) ratio in the right-sided short axis view in early diastole ≥1.6;
- Left ventricular internal diameter in diastole, normalised for body weight (LVIDDN) ≥1.7.
When echocardiography is not available and thoracic radiography is being used to evaluate heart size, a combination of vertebral heart score (VHS) and vertebral left aortic score (VLAS) should be used to reduce subjectivity. This involves measuring the distances between cardiac landmarks on a lateral thoracic radiograph and comparing these to the length of the patient’s thoracic vertebrae. When VHS and VLAS are being used, patients should meet all three of the criteria below to qualify as Stage B2.
- Heart murmur ≥ Grade 3;
- Breed adjusted VHS values ≥ 10.5 or, where a “general” non breed specific value is being used, a value of ≥11.5;
- VLAS score of ≥3.
To measure VHS:
- Obtain a good quality lateral thoracic radiograph centred over the heart.
- Identify the carina – a circular radiolucency dorsal to the base of the heart which corresponds to the bifurcation of the mainstem bronchi.
- Measure the long axis of the cardiac silhouette from the ventral border of the carina to the apex.
- Using the cranial border of the fourth thoracic vertebra as the starting point, express the length of the long axis of the cardiac silhouette in terms of vertebral body units (VBU) to the closest 0.1.
- Next, measure the short axis of the cardiac silhouette from the cranial border to the dorsal border at the widest part, perpendicular to the long axis.
- Express the length of the short axis in terms of VBUs, again using the cranial border of the fourth thoracic vertebra as the starting point.
- Add both numbers together to obtain the patient’s VHS10.
To measure VLAS:
- Use the same good quality lateral thoracic radiograph centred over the heart.
- Identify the caudal vena cava – a thick, soft-tissue opacity vessel running through the caudal thoracic cavity.
- Measure from the ventral aspect of the carina to the dorsal border of the caudal vena cava at the site where it intersects the caudal aspect of the cardiac silhouette.
- Express this distance in terms of VBUs, again using the cranial border of the fourth thoracic vertebra as the starting point11.
The Cardiac Education Group provides a useful interactive tool to gain confidence measuring VHS and VLAS12.
Stage B treatment recommendations
There are currently no pharmaceutical options which have been demonstrated to be helpful in stage B1.
For patients in Stage B2, the EPIC study has shown that pimobendan can slow the progression of DMVD and delay the onset of heart failure13. On average, beginning pimobendan at labelled dose in B2 gave patients an additional 15 months of asymptomatic life before they progressed into Stage C and, therefore, this has now become the treatment recommendation for this population.
Some cardiologists will also administer an ACE inhibitor in Stage B2 but studies evaluating this practice have shown mixed results and so it is more common to reserve ACE inhibitors for Stage C patients14,15,16.
Monitoring patients in Stage B2
When a patient has been identified as being in Stage B2 it is recommended that they should be followed closely to monitor progression of disease. In a study which monitored 135 dogs as they progressed from Stage B2 to Stage C, it was noted that biomarkers such as respiratory rate, resting respiratory rate, VHS and heart rate increased as patients advanced through Stage B2 and into heart failure17. Twelve months before the onset of heart failure, patient VHS increased by 15 per cent (from 11.5 to 13.0); 10 months before the onset of heart failure, heart rate increased by 20 per cent (from 125 bpm to 150 bpm); four months before onset of heart failure, respiratory rate increased by 58 per cent (from 31 to 49); and four months before onset of heart failure, resting respiratory rate as measured by owners at home increased by 78 per cent (from 23 to 41).
Based on these findings, monitoring of B2 patients should include repeat thoracic radiography to track changes in VHS, but where this is cost-prohibitive it is valuable to understand that measuring patient heart rate and respiratory rate can provide an inexpensive proxy. Owners should be taught to measure and record resting respiratory rate at home between clinic visits and should be informed to schedule a recheck if they are consistently measuring average results over 30 breaths per minute. These biomarkers all correlate strongly with advancing disease and can signify when an animal is getting closer to developing clinical heart failure that will necessitate a change in medical treatment. By monitoring patients in this way, it is hoped that we can avoid the situation where a patient with a previously diagnosed heart murmur presents unexpectedly with life threatening pulmonary oedema.
Stage C diagnosis
Dogs who have exhibited clinical signs associated with DMVD are in “congestive heart failure” (CHF) or Stage C. The typical Stage C dog will have signs consistent with pulmonary congestion resulting from left-sided heart failure such as tachypnoea, restlessness, and dyspnoea, or right-sided heart failure such as ascites. Because there is a high incidence of tracheobronchial disease in the same population of small breed dogs which are at risk of developing DMVD, care should be taken to differentiate between cough of cardiac origin and cough originating from tracheobronchial disease. It is not safe to assume that a dog with DMVD which develops a cough is in CHF.
Stage C treatment recommendations
For the patient presenting with pulmonary oedema resulting in severe respiratory distress, oxygen supplementation and diuresis will be priority. Injectable furosemide boluses can be given IV or IM and repeated hourly to alleviate respiratory distress. Patients should have free access to drinking water and monitoring of kidney values and electrolytes should be performed. Identification of kidney disease will present a challenge for long-term management of the cardiac patient. For longer term care, oral loop diuretics like oral furosemide or torasemide may be used and should be tapered to the lowest effective dose. Torasemide is a more potent loop diuretic than furosemide with greater oral bioavailability and a longer half-life meaning that once-daily dosing is often achievable. As patients move through Stage C, diuretic doses will need to be adjusted to keep patients comfortable.
In addition to loop diuretics, there are three other medications which are recommended to be given in combination to dogs in CHF, commonly referred to as “triple therapy”: pimobendan, an ACE inhibitor, and spironolactone. These medications have been shown to improve quality of life and slow progression of heart failure in Stage C patients18,19,20,21. It is worth noting that although spironolactone has weak diuretic properties, its inclusion as standard therapy for dogs in heart failure is not for diuresis, but rather due to its ability to block aldosterone. Chronic renin angiotensin aldosterone system (RAAS) activation results in worsening of CHF and cardiac remodelling, and although ACE inhibitors can block the RAAS cascade, in one study, as many as 32 per cent of dogs with CHF receiving ACE inhibitors experienced aldosterone break through necessitating a multimodal approach to RAAS blockade22. The term “quadruple therapy” is used to describe the standard combination therapy of loop diuretics, pimobendan, ACE inhibitors, and spironolactone for Stage C patients.
Stage D diagnosis
A patient is determined to have moved into Stage D DMVD when they have become refractory to standard therapy for Stage C patients, including “triple therapy” of pimobendan, ACE inhibitors, and spironolactone at standard doses, and when their requirement for oral furosemide has escalated above a total dose of 8mg/kg/day.
Stage D treatment recommendations
Patients who are showing signs of being refractory to oral furosemide may still benefit from injectable furosemide boluses given IV or IM, as congestion and oedema in the gastrointestinal tract as a direct result of CHF can result in reduced oral bioavailability. Switching to oral torasemide, if not already done, may be useful in these patients as this loop diuretic is more potent, has better oral bioavailability, and seems to be less impacted by malabsorption23.
Ultimately, by definition, patients who are refractory to standard therapies will present treatment challenges and, if owners wish to pursue further treatment options, consultation with a cardiologist should again be considered.
Summary
Definitive diagnosis of degenerative mitral valve disease requires echocardiography of the valve leaflets and cordae tendinae, and when echocardiography is available it can be used to stage and monitor subclinical patients by evaluating the level of cardiac remodelling. However, echocardiography may not always be feasible. A contextualised care approach includes making a presumptive diagnosis of DMVD using careful cardiac auscultation and an understanding of the epidemiology of the disease. Murmur grade and thoracic radiographs can be used to stage patients with subclinical heart murmurs, and radiographs and biomarkers can be used to monitor progression of disease. Regardless of which of these paths is taken, the ACVIM consensus guidelines provide evidence-based treatment recommendations for clinicians and a valuable framework around which to discuss disease progression with owners.
- Borgarelli M, Buchanan JW. Historical review, epidemiology and natural history of degenerative mitral valve disease. Journal of Veterinary Cardiology 2012;14:93-101
- Mattin MJ, Boswood A, Church DB, Lopez-Alvarez J et al. Prevalence of and risk factors for degenerative mitral valve disease in dogs attending primary-care veterinary practices in England. Journal of Veterinary Internal Medicine 2015;29:847-854
- Thrusfield MV, Aitken CGG, Darke PGG. Observations on breed and sex in relation to canine heart valve incompetence. Journal of Small Animal Practice 1985;26:709–717
- Pedersen HD, Häggström J, Falk T, Mow T et al. Auscultation in mild mitral regurgitation in dogs: observer variation, effects of physical maneuvers, and agreement with color doppler echocardiography and phonocardiography. Journal of Veterinary Internal Medicine 1999;13:56–64
- Campbell FE. Cardiac disease and examination. World Small Animal Veterinary Association 2013 Congress Proceedings; 2013 Mar 6-9; Auckland, New Zealand. Available from: https://www.vin.com/doc/?id=5709745
- Rishniw M. Murmur grading in humans and animals: past and present. Journal of Veterinary Cardiology 2018;20:223-233
- Häggström J, Kvart C, Hansson K. Heart sounds and murmurs: changes related to severity of chronic valvular disease in the Cavalier King Charles Spaniel. Journal of Veterinary Internal Medicine 1995;9(2):75-85
- Atkins C, Bonagura J, Ettinger S, Fox P et al. Guidelines for the diagnosis and treatment of canine chronic valvular heart disease. Journal of Veterinary Internal Medicine 2009;23:1142–1150
- Keene BW, Atkins CE, Bonagura JD, Fox PR. ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs. Journal of Veterinary Internal Medicine 2019;33:1127–1140
- Buchanan JW, Bücheler J. Vertebral scale system to measure canine heart size in radiographs. Journal of the American Veterinary Medical Association 1995;206(2):194-199
- Malcolm EL, Visser LC, Phillips KL, Johnson LR. Diagnostic value of vertebral left atrial size as determined from thoracic radiographs for assessment of left atrial size in dogs with myxomatous mitral valve disease. Journal of the American Veterinary Medical Association 2018;253(8):1038-1045
- Cardiac Education Group. Interactive radiographic viewer [Internet]. Cardiac Education Group; c2020 [cited 2026 Feb 08]. Available from: https://cardiaceducationgroup.org/learn/interactive-radiographic-viewer/
- Boswood A, Häggstrom J, Gordon SG, Wess G et al. Effect of pimobendan in dogs with preclinical myxomatous mitral valve disease and cardiomegaly: the EPIC study—a randomized clinical trial. Journal of Veterinary Internal Medicine 2016;30:1765-1779
- Kvart C, Häggström J, Pedersen HD, Hansson K et al. Efficacy of enalapril for prevention of congestive heart failure in dogs with myxomatous valve disease and asymptomatic mitral regurgitation. Journal of Veterinary Internal Medicine 2002;16:80-88
- Atkins CE, Keene BW, Brown WA, Coats JR et al. Results of the veterinary enalapril trial to prove reduction in onset of heart failure in dogs chronically treated with enalapril alone for compensated, naturally occurring mitral valve insufficiency. Journal of the American Veterinary Medical Association 2007;231:1061-1069
- Pouchelon JL, Jamet N, Gouni V, Tissier GR et al. Effect of benazepril on survival and cardiac events in dogs with asymptomatic mitral valve disease: a retrospective study of 141 cases. Journal of Veterinary Internal Medicine 2008;22:905-914
- Boswood A, Gordon SG, Häggström J, Vanselow M et al. Temporal changes in clinical and radiographic variables in dogs with preclinical myxomatous mitral valve disease: the EPIC study. Journal of Veterinary Internal Medicine 2020;34:1108–1118
- Bernay F, Bland JM, Häggström J, Baduel L et al. Efficacy of spironolactone on survival in dogs with naturally occurring mitral regurgitation caused by myxomatous mitral valve disease. Journal of Veterinary Internal Medicine 2010;24:331-341
- Häggström J, Boswood A, O’Grady M, Jöns O et al. Effect of pimobendan or benazepril hydrochloride on survival times in dogs with congestive heart failure caused by naturally occurring myxomatous mitral valve disease: the QUEST study. Journal of Veterinary Internal Medicine 2008;22:1124-1135
- Lombard CW, Jons O, Bussadori CM. Clinical efficacy of pimobendan versus benazepril for the treatment of acquired atrioventricular valvular disease in dogs. Journal of American Animal Hospital Association 2006;42(4):249-261
- Häggström J, Boswood A, O’Grady M Jöns O et al. Longitudinal analysis of quality of life, clinical, radiographic, echocardiographic, and laboratory variables in dogs with myxomatous mitral valve disease receiving pimobendan or benazepril: the QUEST study. Journal of Veterinary Internal Medicine 2013;27:1441–1451
- Ames MK, Atkins CE, Eriksson A, Hess AM. Aldosterone breakthrough in dogs with naturally occurring myxomatous mitral valve disease. Journal of Veterinary Cardiology 2017;19:218-227
- Oyama MA, Peddle GD, Reynolds CA, Singletary GE. Use of the loop diuretic torsemide in three dogs with advanced heart failure. Journal of Veterinary Cardiology 2011;13:287-292
1. WHICH OF THE FOLLOWING STATEMENTS IS CORRECT REGARDING THE EPIDEMIOLOGY OF DMVD?
A. Female dogs have an increased risk of DMVD
B. Dogs less than 20kg have an increased risk of DMVD
C. Dogs under the age of five years have an increased risk of DMVD
D. Giant breed dogs have an increased risk of DMVD
2. WHAT GRADE IS A HEART MURMUR WHEN THE INTENSITY OF THE SOUND OF THE HEART MURMUR IS THE SAME INTENSITY AS S1/S2 HEART SOUNDS?
A. Grade 1
B. Grade 2
C. Grade 3
D. Grade 4
E. Grade 5
F. Grade 6
3. A SMALL BREED, MIDDLE-AGED PATIENT WITH A NEWLY DIAGNOSED LEFT APICAL HEART MURMUR BUT NO CLINICAL SIGNS OF CONGESTIVE HEART FAILURE CAN BE PRESUMED TO BE IN WHICH ACVIM STAGE OF DMVD?
A. Stage A
B. Stage B
C. Stage C
D. Stage D
4. WHEN A PATIENT IN ACVIM STAGE B2 DEVELOPS A COUGH, IT IS SAFE TO ASSUME THAT THE PATIENT HAS NOW MOVED INTO STAGE C (CONGESTIVE HEART FAILURE):
A. True
B. False
5. WHAT IS THE EARLIEST ACVIM STAGE FOR WHICH THERE IS EVIDENCE TO SUPPORT MAKING A RECOMMENDATION TO START TREATMENT WITH PIMOBENDAN?
A. Stage A
B. Stage B1
C. Stage B2
D. Stage C
E. Stage D
6. WHICH OF THE MEDICATIONS BELOW IS NOT PART OF STANDARD “QUADRUPLE” THERAPY FOR DOGS IN CONGESTIVE HEART FAILURE?
A. Loop diuretics
B. Pimobendan
C. ACE Inhibitor
D. Spironolactone
E. Amlodipine
ANSWERS: 1B; 2C; 3B; 4B; 5C; 6E.
















