How to Neuter Rabbits
In last month’s SACE article, Head vet at Ark Veterinary Clinics Ltd, exotics lecturer at Glasgow University Veterinary School, and BSAVA Committee Member, Dr Madonna Livingstone BVMS MRCVS discussed why we should be neutering pet rabbits and how to anaesthetise rabbits safely. This month, Dr Livingstone provides readers with an easy to follow, step-by-step guide to the surgical procedures
Ovariohysterectomy in rabbits
My belief with spaying any species is that you make the wound as large as you need to. Wounds heal ‘side to side’ so a four-inch wound will heal as fast as a two-inch one. Most of the vets I see who run into problems peri operatively are trying to operate through a tiny wound.
First, learn to spay correctly, then learn to perform the surgery faster and lastly learn to perform the surgery through a smaller wound. It is completely unrealistic to expect a vet who is two years qualified to spay through a wound the same size as a twenty-year qualified vet. These expectations (sometimes it’s the vet putting pressure on themselves) lead to high stress levels, demoralisation and increased morbidity/mortality.
Fat
The primary fat storage sites in does are the mesometrium and mesovarium. Mature does (six months and older) have significant amounts of fat at these sites even at a normal body condition score (see Figures 1a and 1b). This can make surgery much more difficult as rabbit tissue is very friable compared to more familiar species; when they are overweight, it can result in a stressful procedure with higher risks.
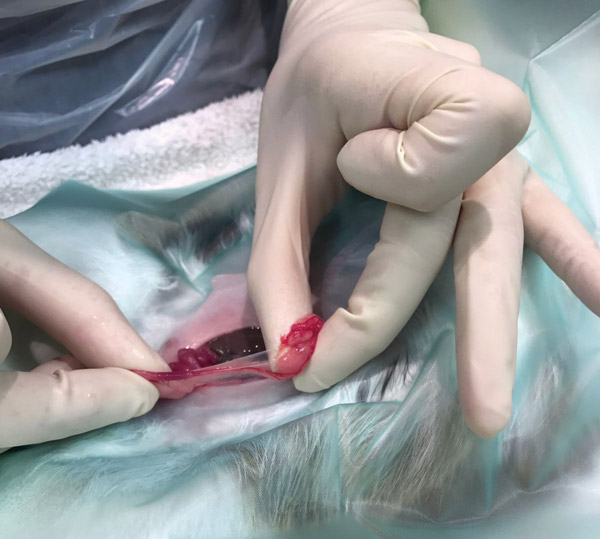
Figure 1a: Rabbit uterus and ovary in a young doe.

Figure 1b: Rabbit uterus and ovary in a mature doe.
Personally, I have a policy of not neutering any animal that is badly overweight. Neutering is an elective procedure, once I have clarified the increased risks associated with operating on an obese patient – I describe the fat melting, highlight the increased risk of bleeding due to everything being slippy and explain how it’s like operating in a chip pan – the vast majority of owners ‘get it’.
Neutering obese animals is, in my opinion, not good animal welfare and I advise my owners that I will always put their animal first. If owners cannot correct their husbandry pre-op they will not do it post-op. I do not shame owners, education and empathy is key to getting them on board. Since instigating this policy in the practice fifteen years ago, we have only had one complaint about the policy, and our vets do not suffer the stress of spaying fat bitches/cats/rabbits.
I recommend neutering rabbits at fourteen to sixteen weeks of age. Giant breeds take longer to mature and I will generally leave the giant breeds until six months of age before recommending neutering.
Species Specific Considerations
Apart from anaesthesia considerations, and the friability of rabbit tissue, there are five main differences between spaying rabbits and spaying more familiar species:
Rabbits have a large caecum in close proximity to the abdominal wall; tenting the linea alba upwards is important on entry to the abdomen to prevent inadvertent piercing of the caecum.
Rabbits have no uterine body. There are two horns, two cervixes and the structure which is often mistaken for a uterine body is in fact the flaccid internal vagina. The internal vagina frequently fills with urine when the doe urinates.
The ovaries are approximately an inch away (sometimes much more) from the end of the uterine horns. It is important to appreciate this and visualise the ovaries as there have been several cases where the ovaries have been left behind (see Figures 1a and 1b).
Catgut should not be used. Rabbits struggle to break catgut down leading to increased risks of adhesions and granuloma formation which can cause chronic pain.
Rabbits form adhesions readily and are used in human adhesion studies because of this. In humans, abdominal adhesions are extremely painful. Gentle tissue handling will help reduce the risk. If swabs are required in the abdomen, then pre-soaking them with saline will reduce the risk of drying out viscera. If any of the viscera or fat becomes dry, it will increase the risk of adhesions. Flushing the abdomen with warmed sterile saline post-operatively can also help reduce the risk and will hydrate your patient.
How to Perform a Rabbit Ovariohysterectomy
The umbilicus can be difficult to appreciate in many rabbits. An easier landmark is the caudal pair of nipples (see Figure 2). Holding your scalpel in a pen-like grip, make a skin incision midline approximately two inches long, between the nipples.
Blunt dissect through the subcutaneous layer to expose the muscle. The thickness of the subcutaneous layer varies between individuals and in some it is almost absent. The linea alba is sometimes not as obvious as we are used to in dogs. There are often three white lines running roughly parallel – the middle one is the linea alba.

Figure 2: Landmarks for initial incision. The umbilicus is highlighted by the blue arrow.
Grasp the linea alba in your rat tooth forceps (ensuring you have only grasped the muscle and not underlying viscera), tent the muscles up as high as you are comfortable with (the muscle layer is not overly friable) and perform a reverse stab incision (see Figure 3). We tent the muscle to elevate away from the large caecum. Enlarge the opening in the muscles using scissors, ensuring the tips are pointing up (so we know what we are cutting).
The uterus sits between the bladder and the descending colon. Locate the end of the uterine horn, the infundibulum will run cranially from the end of the horn and then circle back around the ovary. If the ovary does not exteriorise easily, massage the fat distal to the ovary between your fingers which will often loosen the tissue. The ovarian artery runs perpendicular to the ovary (see Figure 4). Visualisation of the artery is not always possible due to fat storage. An avascular window is present cranial and caudal to the ovary (see Figure 5).
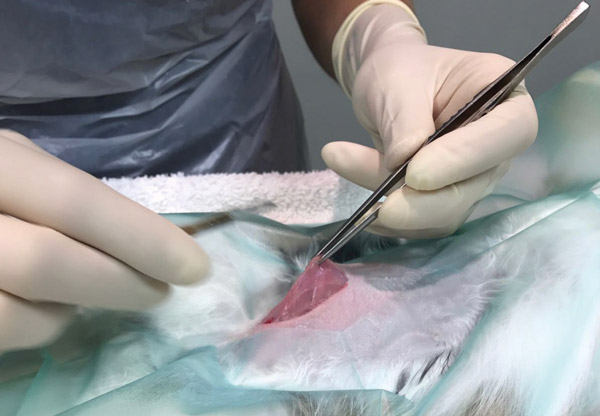
Figure 3: Rabbit muscle tenting for surgery.
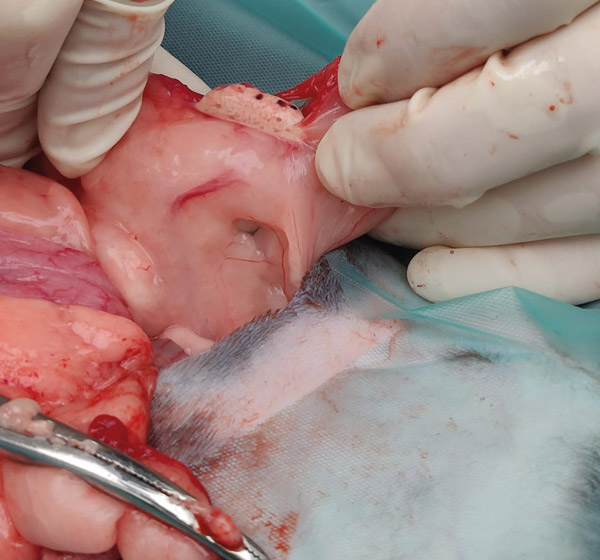
Figure 4: Ovarian artery.
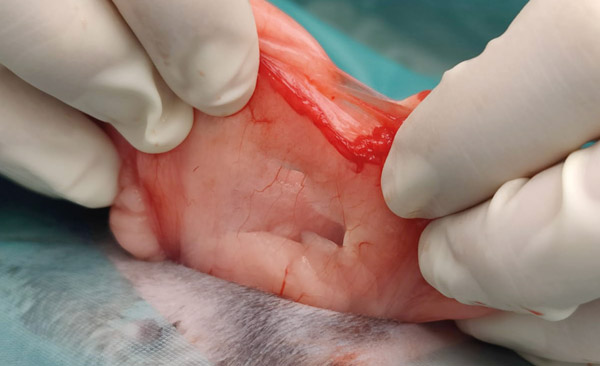
Figure 5: Avascular window beside ovary.
Holding the ovary in your non-dominant hand, break down the avascular windows to allow placement of artery forceps across the ovarian artery, or where you know it will be (see Figure 6). Do not pre-crush the ligature site. Using 3metric Vicryl or similar material place two ligatures. Place another artery forcep directly above the one already placed, so that the two are touching. Keeping the one closest to the ligatures still and steady, rotate and twist the other forcep away (see Figure 7). This causes the tissues to stretch and tear; any remaining tissue can be cut.
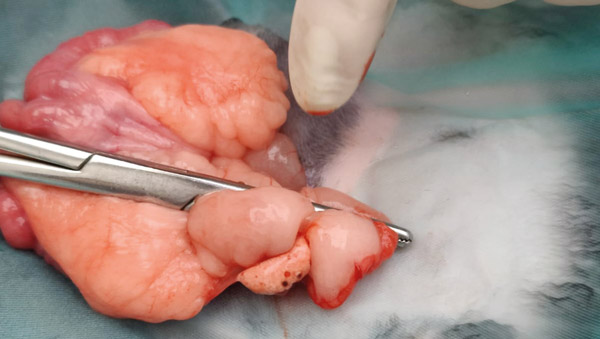
Figure 6: Clamp placed on ovarian artery.
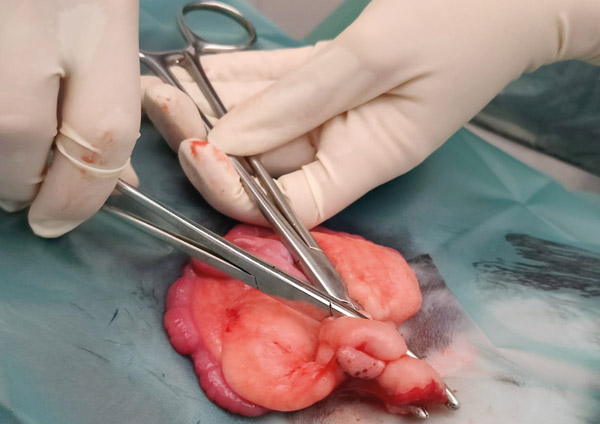
Figure 7: Holding bottom clamp still.
Check for any bleeding before letting go of your ovarian stump. I leave approximately 1cm tails on my ligatures, this allows me to find the site quickly if I am worried about a bleeding stump.
The uterine artery runs parallel to the internal vagina and past the cervixes. Approximately a third of the way along the uterine horn it forms multiple branches. Ligating before it branches is much easier (see Figure 8). Do not place the ligatures too near the internal vagina as there is a risk of incorporating the ureter. Massage the mesometrium to find the avascular window on either side of the uterine artery. Place your artery forcep through these windows and across the artery. Do not pre crush the ligature site. Using 3 metric Vicryl or similar material place two ligatures. Place another artery forcep directly above the one already placed, so that the two are touching. Keeping the one closest to the ligatures still and steady, rotate and twist the other forcep away. This causes the tissues to stretch and tear, any remaining tissue can be cut. Check for any bleeding before letting go.
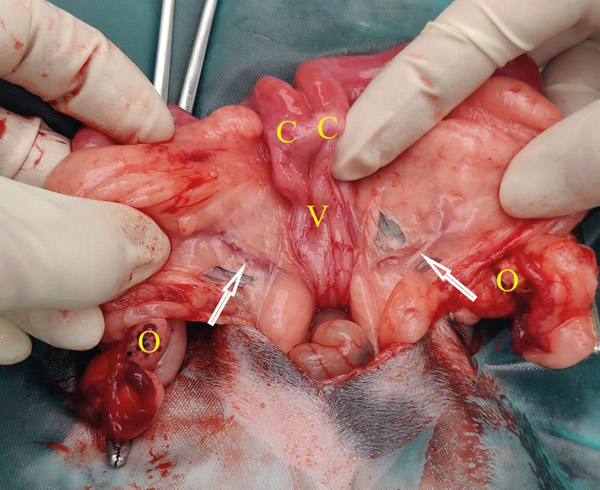
Figure 8: Uterine anatomy doe. The white arrows indicate the uterine arteries before they branch. The ovaries, cervixes and internal flaccid vagina are marked with their respective letters.
How to remove the uterine horns is much debated. Ligating above (uterine side), through and below (internal vaginal side) the cervixes are all recognised techniques with their own pros and cons. My preference is through or just below the cervix. Ligating above the cervixes will inevitably leave some uterine tissue behind. In very young does, this is unlikely to cause a problem as the ovaries have been removed before the uterus has been exposed to higher levels of sex hormones. In older does, there is a risk of uterine adenocarcinomas forming if uterine tissue is left behind. As the internal flaccid vagina will sometimes flood with urine during urination, ligating below the cervixes carries a theoretical risk of uroabdomen if the stump is not oversewn.
Place one artery forcep across both cervixes; using 3/0 Vicryl on a needle, place a transfixing ligature between the cervixes. I then place a circumferential ligature distal to the transfixing.
Place another artery forcep directly above the one already placed, so that the two are touching (see Figure 9). Keeping the one closest to the ligatures still and steady, rotate and twist the other forcep away. This causes the tissues to stretch and tear, any remaining tissue can be cut. Check for any bleeding before letting go.
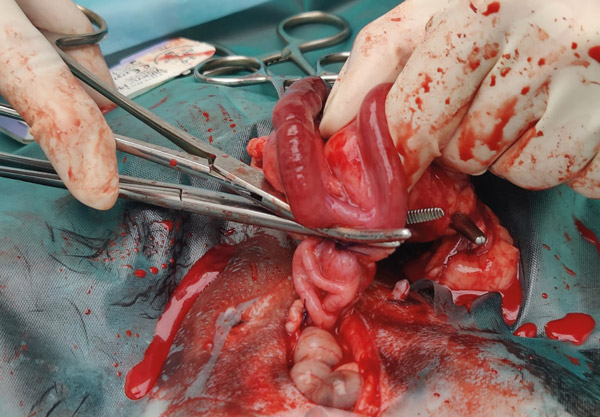
Figure 9: Placing second clamp at cervixes. The fluid is mainly melted fat and a little blood.
Check all stumps for any signs of bleeding then flush the abdomen with warmed sterile saline. Any bleeding stumps should be religated.
Collaclot is an avian collagen sponge (available from IMS wholesalers) which can stop a life-threatening bleed. It is extremely effective and much less expensive than other collagen sponges on the market. They have a long shelf-life and can be used in any species.
Repair the muscle layer with cruciate sutures (3/0 Vicryl on a needle), lifting the muscle to see placement of the needle at all times (I have seen students stitch the muscle onto the caecum when operating on cadavers) (see Figure 10). Rabbits can move unpredictably in multiple directions (think binky, i.e., when a rabbit jumps straight up and quickly twists its hind end, head, or both); cruciate sutures counteract tension in every direction, they are faster to place and have few knots.
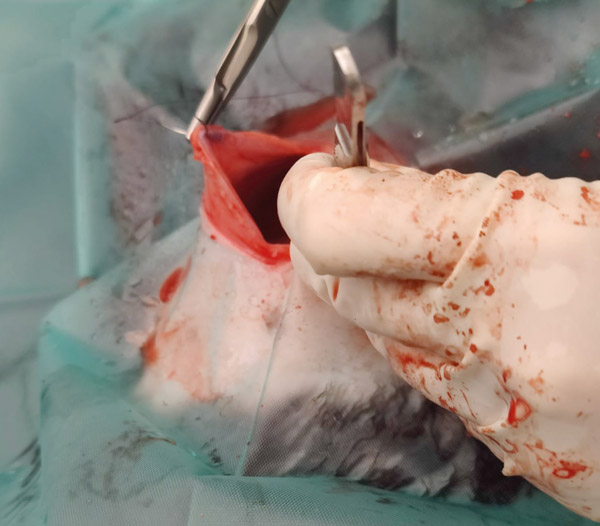
Figure 10: Sutures in abdominal muscles.
Further analgesia can be provided by performing local anaesthesia splash blocks on closure. A mixture of Lidocaine 2mg/kg and Bupivacaine 1mg/kg can be used. Doses higher than this may lead to toxicity.
If there is a subcutaneous layer, this can be closed (4/0 Vicryl on a needle) using a simple continuous pattern, then an intradermal pattern for skin closure. Buster collars should not be used on rabbits – they create stress (which leads to anorexia) and prevents eating of caecotrophs. The majority of rabbits will not interfere with their wounds if they have adequate analgesia on board, which should be continued for ten days. Restrict exercise post-operatively for a week.
Castration
Castrating rabbits is essentially the same procedure we do regularly in other species – we remove the testicles and want a live patient at the end of it.
Species Specific Considerations
Apart from the anaesthetic considerations and avoiding the use of cat gut, the only other difference is that rabbits have open inguinal canals, therefore if we do not close the tunic, our patient has a real risk of abdominal organs herniating into the vaginal tunic/scrotum with potentially devastating consequences. The open inguinal canal can also make castration challenging if we do not stabilise the testicle as the slightest pressure exerted on the testicle will push it back into the abdomen. If this happens, gentle pressure just cranial to the pelvis will usually result in the testis slipping back into the scrotum.
The two techniques I teach are ‘open/closed’ or ‘closed’ castrations. When I first started castrating rabbits, the open/closed technique gave me the confidence that I had indeed taken all of the testicle. Now I am more confident, the closed technique is faster. I hold my scalpel in a pen-like grip as this gives me much better control of the direction and depth of my incision.
Basic Castration Technique
Hold the base of the testicle in your non-dominant hand, clamped between your middle finger and ring finger; the testicle should now be on the palm side of your hand (See Picture 11a). Stabilise the testicle between your thumb and middle finger. Use your index/pointer finger to stretch the scrotal skin. Do not let go of the base of the testicle. The scrotal skin is fragile in rabbits, so not much pressure is required to incise through it. Excess pressure will result in going through the skin and straight through the vaginal tunic and potentially into the testicle.
Holding the scalpel in your dominant hand, feather light stroke in the centre of the scrotal sac, extending roughly half the length of the testis (see Figure 11b).
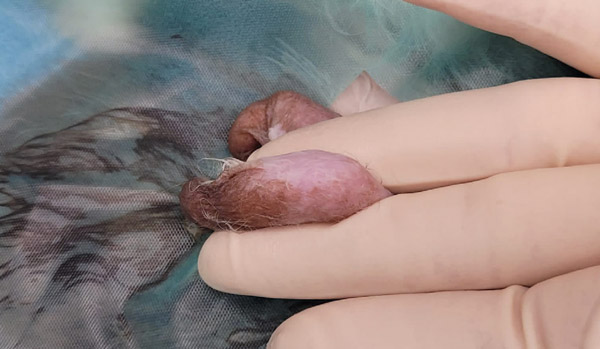
Figure 11a: Holding the testicle to prevent slipping into the abdomen.
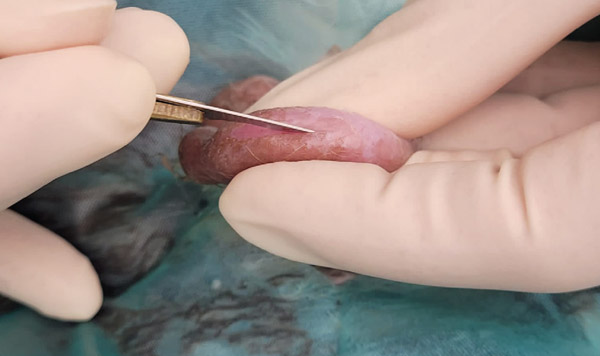
Figure 11b: Sutures in abdominal muscles.
Using blunt-blunt small scissors, push between the scrotal skin and vaginal tunic then open the scissors. This utilises blunt dissection to break down the attachments between the tunic and the scrotum. Repeat in all four quadrants (see Figure 12). Using blunt-blunt scissors ensures we do not accidentally pierce the testicle.
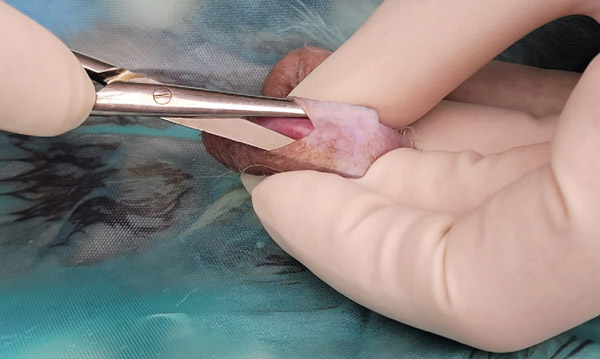
Figure 12: Blunt dissecting between scrotum and vaginal tunic.
Use your index finger and thumb of your non-dominant hand to gently squeeze the testicle caudally while your index finger and thumb of your dominant hand squeeze the caudal pole (epididymis) of the testicle cranially (see Figure 13). This results in the testicle (which is still within the vaginal tunic) popping up through your incision. Once the epididymis is visualised through the tunic, we place an artery forcep across the epididymis (see Figure 14). Now the testicle is stabilised by the artery forcep, we can let go with our non-dominant hand. Hold the artery forcep in your non-dominant hand (do not pull upward) and use a swab to help strip the rest of the scrotum down off the tunic. The tissue can be quite tough in mature bucks.

Figure 13: Squeezing either pole of the testicle to pop it through the scrotal sac.

Figure 14: Place artery forcep on the epididymus and break the attachment between the vaginal tunic and the scrotal sac.
Open/Closed Technique
Mature bucks often have very thick tunics and the testicle is not always easily visualised. Rabbits also have a very elongated testis and have a long fat pad (see Figure 15). This means it can be difficult for inexperienced rabbit surgeons to be confident they have the entire testicle. Making a small incision into the vaginal tunic directly over the testicle and milking the testicle through the incision allows direct visualisation and confirmation that the whole testicle will be taken. This is the ‘open’ part of the technique.

Figure 15: Rabbits have an elongated testis and a long fat pad.
Place artery forceps across the tunic 1cm cranial to the base of the testis. My personal ligature of choice is Vicryl or a similar material. I do not pre-crush ligature sites in rabbits as the tissue can be very friable. 3/0 Vicryl on a needle is used to perform a transfixing ligature which includes the tunic, vascular bundle of the testis and the vas deferens. I also place a circumferential ligature (my preference in all species is to place two ligatures if I have space). This converts the technique back into a ‘closed’ technique. No viscera can herniate as the tunic is now closed.
Place another artery forcep directly above the one already placed, so that the two are touching. Keeping the one closest to the ligatures still and steady, rotate and twist the other forcep away (see Figure 16). This causes the tissues to stretch and tear, any remaining tissue can be cut. Check for any bleeding and tuck the stump back into the scrotal sac.
Wound glue is used to close the scrotal sac (see Figure 17).
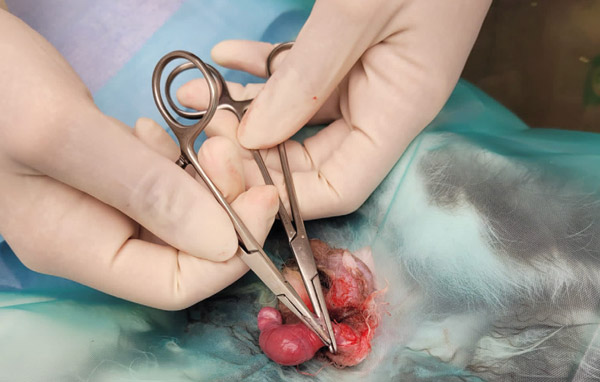
Figure 16: Removing the testicle.

Figure 17: Hold the edges of the scrotal sac in alignment before applying the tissue glue.
Closed Technique
The only difference is that we skip the step of opening into the tunic to check we have the full testicle.
It is very common for the penis to protrude during surgery which is not concerning. Syringe feed on recovery and send home with a week of meloxicam.
Conclusion
Rabbits are popular, much beloved pets and can easily be incorporated into any practice with a bit of lateral thinking. Gentle tissue handling, careful ligature applications (ensure all movement comes from the wrist and do not tug up on the tissues when tying the ligatures), and good multimodal analgesia should be our aim in any surgical procedure in any species.
1. It is important to close the vaginal tunic during castration because:
A. We need to minimise the risk of infection leading to peritonitis
B. Rabbits have closed inguinal canals
C. Rabbits have open inguinal canals
D. Rabbits have delicate tissue
2. True or false: The ovary in rabbits is in the same position as in more familiar species
A. True, the ovary is directly at the end of the uterine horn
B. False, the ovary is approximately an inch or so cranial to the end of the uterine horn
C. True, the ovary is within the vaginal tunic
D. False, the ovary is approximately an inch or so
caudal to the end of the uterine horn
3. The internal female rabbit reproductive tract consists of:
A. Internal flaccid vagina, two cervix, two uterine horns, two infundibulum, and two ovaries
B. Internal flaccid vagina, two cervix, two uterine bodies, two uterine horns, two infundibulum, and two ovaries
C. Two cervix, two uterine bodies, two uterine horns, two infundibulum, and two ovaries
D. One cervix, one uterine body, two uterine horns, two infundibulum, and two ovaries
4. Rabbits form adhesions very easily. How can we minimise this?
A. Gentle tissue handling, use catgut, wet swabs and warm sterile saline to flush abdomen before closing
B. Gentle tissue handling, do not use catgut, wet swabs and warm sterile saline to flush abdomen before closing
C. Gentle tissue handling, use thickest suture available, wet swabs and warm sterile saline to flush abdomen before closing
D. Gentle tissue handling, do not use catgut, dry swabs and warm sterile saline to flush abdomen before closing.
ANSWERS: 1C; 2B; 3A; 4B.
















