Canine alimentary lymphoma: clinical presentation and treatment options
Susana Martinez Valls BVetSc, MRCVS Hamilton Specialist Referral, and Katarzyna Purzycka DVM, MVetMed, MRCVS, DipACVIM (Oncology), Lumbry Park Veterinary Specialists, review the current literature on canine alimentary lymphoma (AL) with an emphasis on clinical behaviour and treatment
Lymphoma is the most common haematopoietic cancer reported in dogs, and the gastrointestinal tract (GIT) is the most frequently involved extra nodal site. The most common clinical presentation is multicentric lymphoma with concurrent involvement of gastrointestinal tract (GIT) reported in some cases. The primary AL (also known as gastrointestinal lymphoma) accounts for five to seven per cent of all canine lymphoma cases. It arises from the GIT, typically without involvement of peripheral lymph nodes (LNs). This, in particular, can contribute to delay in diagnosis and treatment. Although the prognosis for most cases is guarded, more recently, few distinct anatomical forms of AL with more favourable outcomes have been recognised.
CLINICAL PRESENTATION
Alimentary lymphoma can affect dogs of any age although typically adult dogs (median age of nine) have been most commonly reported. No clear breed predisposition exists, although breeds reported to have a higher incidence include Boxers, Crossed breeds, Labrador Retrievers, Pugs, Shar Peis, Golden Retrievers, and Rottweilers. Few studies also suggested male dogs as predisposed.
Dogs with alimentary lymphoma present most commonly with gastrointestinal signs such as vomiting, diarrhoea, hyporexia or anorexia, weight loss, melaena, haematochezia and tenesmus.
Bloodwork abnormalities are usually non-specific. The most commonly reported anaemia is followed by neutrophilia and increased hepatic enzyme activities. Hypoalbuminemia is reported to be the most common finding in dogs with alimentary lymphoma, being present in 50 per cent in Lane et al (2018), 55 per cent of dogs in Sogame et al (2018), 61 per cent of dogs in Rassinik et al (2009) and in 69 per cent of dogs in Couto et al (2018).
The most common anatomical sites for alimentary lymphoma are the small intestine, followed by the stomach and large intestine. Mesenteric lymph nodes, spleen, and liver may be involved as well.
IMAGING FINDINGS AND DIAGNOSIS
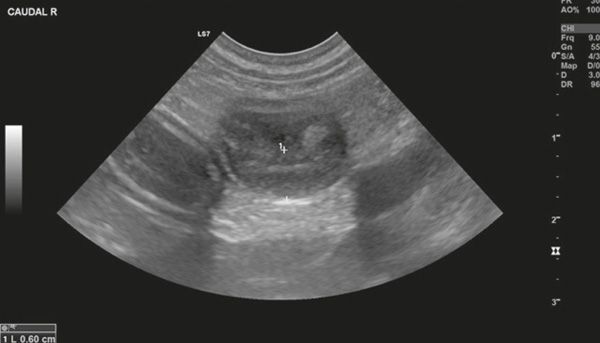
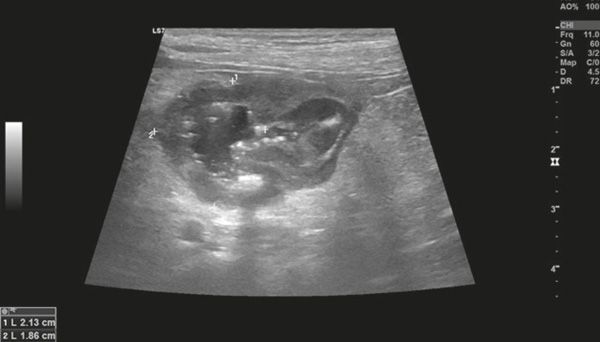
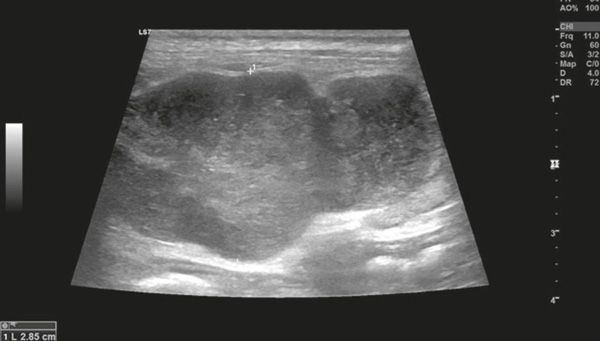
Diagnostic imaging forms a crucial part of clinical workup. Abdominal radiography might not be helpful as the changes in the gastrointestinal tract associated with AL depend on the disease severity, but we can observe a loss of detail or mass effect either due to the gastrointestinal mass and/or lymphadenomegaly or, less often, due to free abdominal fluid. Abdominal ultrasound is more commonly used. The most frequent ultrasonographic abnormality observed is abnormal wall layering with or without concurrent increase in wall thickness (Figure 1). The extent of wall changes can be highly variable in dogs with intestinal lymphoma. Where sonographic abnormalities were present, lesions have been characterised as: predominantly hypoechoic, focal or multifocal ulcerations, small hypoechoic nodules to larger masses (Figure 2) and enlarged abdominal lymph nodes (Figure 3). In some cases abdominal effusion may be present as well. The sonographic appearance of gastrointestinal lymphoma is variable and can overlap with that of a normal bowel, especially in cases of small cell or low-grade AL. Lastly, diffuse intestinal thickening has also been reported and it should be differentiated from enteritis. It is important to note that even when no ultrasonographical abnormalities are found, AL cannot be ruled out without obtaining biopsies of the gastrointestinal tract.
The diagnosis of lymphoma can be made on histopathology where the biopsies are obtained via exploratory laparotomy (resection and anastomosis of a mass or full-thickness intestinal biopsy), upper and/or lower endoscopy (partial-thickness intestinal biopsy); or cytology of ultrasound guided percutaneous fine needle biopsy. Full-thickness biopsies have been demonstrated to be superior in reaching a definitive diagnosis compared to endoscopy biopsies, as neoplastic cell population may be located deeper in the intestinal wall and may be overlooked if only mucosa or submucosa are examined in one study. In 3/10 dogs with low-grade AL undergoing laparotomy in that study, only samples from the ileum were confirmatory for lymphoma, suggesting that ileal sampling might be critical in dogs with suspected AL. Histopathological diagnosis of AL, in particular low-grade or small cell might be challenging based on a routine hematoxylin and eosin (HE) stain, as chronic enteropathies (such as lymphoplasmacytic enteritis) might have a similar morphology. Further testing including immunohistochemistry, flow cytometry and/or PARR might be beneficial and often necessary to reach a final diagnosis.
Characterising the immunophenotype not only aids in achieving a diagnosis but might also guide treatment decision-making and prognosis. The most commonly used markers are: CD3, CD4/CD8 as T-cell markers or CD20, CD79a as a B-cell marker. Other markers used for B-cell lymphoma are CD79a. In terms of cellular morphology, AL is divided into small cell (typically low-grade), intermediate and large cell (typically high-grade). High-grade AL with T-cell immunophenotype is the most commonly diagnosed form. Low-grade alimentary T-cell lymphoma is the most common form of AL in cats and it was believed it was extremely rare in dogs. However, its clinical often indolent course has been recently described in two studies.
Colorectal lymphoma in dogs is typically high grade and B-cell origin (Table 1).

Table 1: Prevalence of B-cell and T-cell immunophenotype in canine AL.
TREATMENT AND PROGNOSIS
The use of a multi-agent chemotherapy protocol is considered the treatment of choice for AL in dogs. Unfortunately, due to the rapid clinical course and aggressive nature of high-grade AL, a long-term prognosis is guarded even with multi-agent chemotherapy protocols with reported median survival time of 62 and 77 days. Several protocols have been investigated (multi agent CHOP- or COP-based chemotherapy protocols, MOP, MVPP, LOPP and single agents [CCNU or L-asparginase]) and high-grade AL still remain a therapeutical challenge. Recently identified increased expression levels of MDA-1 RNA in T-cell AL (similar to the literature describing multicentric lymphoma) suggested that perhaps the use of alkylating agents in the treatment regime might lead to a better outcome. There are still no studies to strongly support it.
Histopathological assessment along with immunophenotyping can guide treatment and prognosis in dogs with small cell T-cell intestinal lymphoma. It has been reported to have a more favourable outcome when undergoing any type of therapy, with a prolonged survival (median survival time of 628 days) compared with historic reports of dogs with large cell, high grade GI lymphoma.More specifically, a combination of steroids and an alkylating agent such as chlorambucil resulted in prolonged survival in this group of dogs. Additionally, dogs with AL localised to large intestine have been shown to have a prolonged survival, which was subsequently reported in a larger case series by Desmas et al (2016). In this study, dogs with colorectal lymphoma treated with a multi-agent chemotherapy had a median survival time of 1,697 days. Surgery is not routinely performed, unless it is necessary to obtain a diagnosis and to treat intestinal perforation or partial/complete obstructions secondary to solitary lesions.
There are prognostic factors identified. One of them is haematochezia in dogs with colorectal AL, which has been associated with a positive impact on the progression-free survival (PFS). This might be because it is more noticeable and alarming for the owners so it leads to earlier diagnosis and treatment. Other gastrointestinal clinical signs were reported to be negative prognostic factors at the time of diagnosis, possibly as a result of a more advanced disease. In addition, Couto et al (2018) reported that dogs with anaemia, weight loss and a low body condition score at the time of diagnosis of small cell, T-cell AL had shorter survival, possibly as a result of more severe infiltration resulting in weight loss (secondary to malabsorption). Anaemia at the time of diagnosis might be related to other co-morbidities such as inflammatory bowel disease, iron deficiency anaemia related to chronic gastrointestinal haemorrhage, or production of anaemia inducing substance by tumour cells. In Rassnick et al (2009), diarrhoea and a lack of response to chemotherapy was noted to be a negative prognostic factor. Hypoalbuminaemia is thought to be secondary to the loss of low molecular weight proteins across the compromised intestinal wall and it is also thought that the presence of this abnormality does not influence survival. This is in agreement with Lane et al (2017) who describe no significant association between PFS or median survival time in dogs and hypoalbuminaemia. Finally, the reduction in the absorption of cobalamin in the ileum can result in hypocobalaminaemia which has been shown to be a negative prognostic factor.
CONCLUSIONS
Although AL in dogs is less common compared to multicentric lymphoma, it is clinically relevant. Its diagnosis might involve additional tests since different subtypes have different treatment and prognosis.
NOTE: Part Two of the waterfowl management and care article by Ana Vale DVM, MVS, PhD, Assistant Professor in Veterinary Public Health, UCD, which featured in SACE in our February edition, will follow in mid-summer.
1. Which of the following sites are most commonly affected in dogs with alimentary lymphoma:
A. Mesenteric lymph nodes and spleen
B. Liver and pancreas
C. Small intestine and stomach
D. All of the above
2. Which of the following best describes achieving a diagnosis in dogs with alimentary lymphoma?
A. Dogs with alimentary lymphoma had commonly bone marrow involvement, therefore a diagnosis can be achieved via bone marrow biopsy
B. Partial thickness biopsies are preferred over full thickness biopsies
C. An additional ileum sample has been proposed as an important site of sampling for confirmation of AL
D. Diagnosis of AL can be reached after abdominal ultrasound only
3. What is the current treatment of choice for canine alimentary lymphoma?
A. Surgery followed by chemotherapy
B. Surgical resection of the lesion followed by active surveillance
C. Multiagent chemotherapy
D. Single agent chemotherapy
4. Which of the following has been identified as a positive prognostic factor in canine alimentary lymphoma?
A. Diarrhoea
B. Haematochezia
C. Anorexia
D. Hypocobalaminaemia
5. Which of the following clinical presentations of alimentary lymphoma have better outcomes?
A. Colorectal lymphoma
B. Low grade lymphoma
C. Gastric lymphoma
D. A and B are correct
Answers: 1C; 2C; 3C; 4B; 5D.