Coccidiosis in ruminants: the inevitable infection
Sarah Campbell MVB Cert DHH MRCVS, veterinary adviser, MSD Animal Health, explores coccidiosis in ruminants, its diagnosis, control and the pharmacological options available
Infection of ruminants by pathogenic Eimeria species is inevitable.1 Oocysts are ubiquitous in the environment where animals are kept, and are capable of surviving for long periods of time, thus, all calves become infected sooner rather than later.1 The risk of infection is increased where stocking density is high or faecal material accumulates coinciding with optimal environmental conditions for oocyst survival. The risk of succumbing to disease is increased when infected animals are exposed to high infectious pressure and management stressors at the same time.2
The prevalence of Eimeria infection in cattle is high and can reach 100% in calves.1,3,4,5 Whether an animal develops clinical signs or is affected sub-clinically depends on the infectious dose, pathogenicity of species involved and the degree of concurrent stress factors prevailing.
Coccidiosis is caused by the protozoan parasite of the genus Eimeria and nearly all vertebrate animals host one or more species. Thousands of coccidian species have been described although many are non-pathogenic. Eimeria are host-specific and mixed infections with more than one species are more common than single-species disease.2,5 There are around 13 species of significance in cattle of which Eimeria bovis, Eimeria zuernii and Eimeria alabamensis are the most important. In sheep, there is a similar number of pathogenic species although Eimeria ovinoidalis, Eimeria bakuensis and Eimeria crandallis are responsible for most disease.
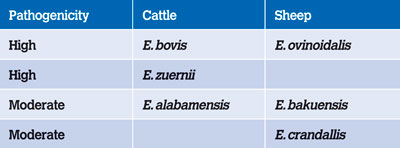
Table 1: Pathogenicity of Eimeria species of cattle and sheep.5
Economic impact
The economic impact of coccidiosis infection is estimated to be high on Irish beef, dairy and sheep farms. The outcome of infection with Eimeria species can lead to partial species-specific immunity while disease can be either clinical or subclinical. Losses associated with clinical cases are obvious and result from deaths, the cost of treating clinical cases along with an increased susceptibility to secondary infection and subsequent disease. In contrast, subclinical losses are often less apparent. These result from reduced feed-conversion efficiency and higher production costs due to delayed time to reach weight targets.1 Considering that coccidiosis can have a negative impact on growth and performance of young animals, it remains a disease of significant economic importance.
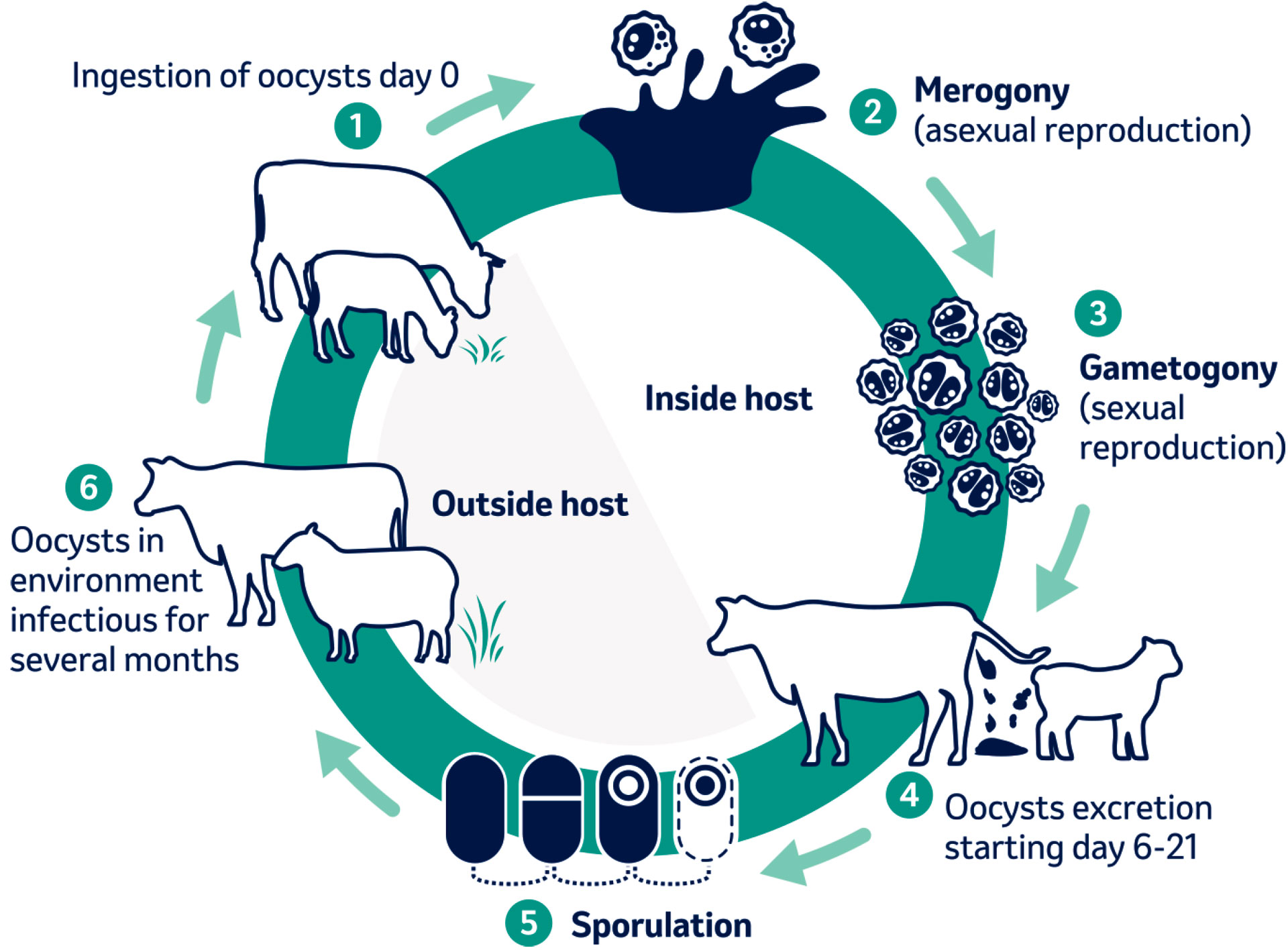
Figure 1. Life cycle of coccidiosis.
Life cycle
The life cycle can be divided into two phases: an exogenous phase (environmental stage) and an endogenous phase (within the host). The length of the life cycle varies depending on the species but usually takes between two to four weeks to complete.6 The prepatent period (time from initial infection to onset of oocyst shedding) and the patent period (duration of oocyst shedding) varies depending on the species. Oocyst excretion can be as short as one day for E. alabamensis
During the exogenous phase, unsporulated oocysts that were excreted in faeces undergo sporulation under favourable environmental conditions such as moist, mild areas in the presence of oxygen. Sporulation results in infective oocysts within days but can take longer in extremely dry, hot or very cold environments.5
Upon ingestion of infective-stage sporulated oocysts, excystation occurs releasing sporozoites into the gastrointestinal tract of the host. Sporozoites invade the nearby small intestinal cells and transform into schizonts and multiply asexually by a process known as merogony. Merozoites are released and re-infect the host intestinal mucosal cells further along the gastrointestinal tract resulting in additional damage and destruction. Multiple phases of asexual reproduction can occur with the final stage of sexual reproduction (gametogony) producing unsporulated oocysts which pass out in faeces. The severe damage to the gut lining caused during sexual reproduction of the parasite contributes greatly to clinical signs observed.6
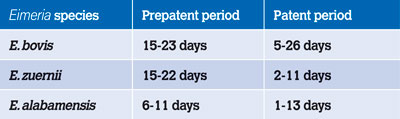
Table 2: Length of prepatent and patent period of bovine Eimeria species.1
Epidemiology
Coccidiosis is a group disease rather than an individual animal issue. The correlation between stress and the onset of clinical coccidiosis is believed to enhance the risk of an outbreak. An entire group is usually exposed to a similar burden of Eimeria infection in the same environment and are subjected to the same external stressors.1 Therefore, individual animal treatment is suboptimal and the whole group should be considered infected.
Oocysts are usually present in soil, vegetation or water sources of all sites inhabited by ruminants. While older animals act as a reservoir for disease, young animals can contribute to high levels of environmental contamination and are the main source of amplification and spread to cohorts. Most animals greater than one year old develop partial immunity to infection after adequate exposure and excrete small numbers of oocysts. A periparturient rise in Eimeria oocysts may occur in cows and sheep.
Coccidia are highly prolific. Each sporulated oocyst can produce 23 million oocysts during the endogenous phase of replication.6 This leads to high levels of build-up in the environment, especially where animals are likely to defaecate such as around feeding troughs, water drinkers as well as outdoors on overstocked paddocks.1 Once Eimeria species are present, the parasite spreads extensively through the herd within a few life cycles.
Oocysts are extremely resistant in the environment and can remain viable for at least one year. Due to their thick wall, they are capable of withstanding long periods of low oxygen availability, extreme pH changes, high temperatures and freezing.2,5 Uninterrupted periods of high temperatures (greater than 39oC) can prevent oocyst sporulation although this is unlikely to occur in the Irish climate.7 Mild, moist conditions favour sporulation. Covered areas that are wet and contaminated by faecal matter are associated with an increased risk of infection.2
Direct exposure to ultraviolet light and extreme dryness is harmful to oocyst survival. Therefore, good hygiene along with exposure of animal equipment and housing to direct sunlight can reduce the environmental burden.5
In combination with high infectious pressure, the presence of stressors such as mixing, weaning, overcrowding and poor nutrition, facilitates the development of clinical coccidiosis in host animals. The main impact of coccidial infection results from subclinical disease. Affected groups may have lower live weight gain and reduced feed-conversion efficiency due to the presence of intestinal lesions.
The species that commonly cause disease in cattle vary in pathogenicity. Very high doses of oocysts are needed to induce experimental clinical disease in calves with
E. alabamensis. In comparison, experimental trickle infections with quite low doses of 100 oocysts of E. bovis daily for 48 days could induce clinical disease in calves.1
E. alabamensis can be detected in calves within the first two weeks of turnout. The species can occur during the housing period although clinical disease is more often seen at pasture and the prevalence is generally low in young calves.8 Along with extremely high doses of oocysts needed to cause disease, it has been suspected that stress and a sudden change in diet associated with turnout may increase the risk of disease occurring. Grazing the same paddocks repeatedly with young stock can contribute to a build-up of infective oocysts on pasture from one season to the next leading to disease in naïve calves in the absence of predisposing factors.1
Immunity
Exposure to pathogenic Eimeria species is required for immunity to develop. Most animals are immune by one year old. The degree of immunity that develops depends on the quantity of oocysts picked up during the primary infection.1,5 A low dose may not deliver the antigenic stimulus necessary to trigger the immune response enough to prevent replication and disease.1 Immunity is usually partial and strictly species-specific. Partial immunity is protective against the clinical manifestation of disease but does not supress parasite replication completely upon subsequent exposure.2 Cross protection between species does not occur therefore it is possible for animals to be prone to coccidial infection more than once. The majority of infections in ruminants involve more than one species at a time rendering multiple disease episodes unlikely.
Acquired immunity develops rapidly and is maintained by continuous exposure. It is mainly based on a cellular immune response involving T helper cell 1 (Th1). A humoral response also occurs with the formation of specific antibodies but is unlikely to contribute to protection.1,5
Pathophysiology
Due to the intracellular nature of parasite development, severe harm occurs upon eruption from intestinal cells during asexual and sexual replication. Several factors influence the degree of damage such as the infective dose, the host immune inflammatory response and simultaneous infection with other gastrointestinal pathogens.5
Cattle
Calves between three weeks’ and six months’ are most susceptible to the effects of disease but shedding can occur transiently throughout life in low numbers.5
The initial site of invasion for most Eimeria species is the small intestine.5 Lesions can persist long after parasite replication has ceased, leading to prolonged effects on intestinal function. Most damage to intestinal lining occurs during later asexual (schizogony) and sexual (gametogony) stages of replication. Invasion of deeper layers of the intestinal wall by E. bovis leads to even greater pathology.
In cattle, extensive epithelial destruction in the distal small intestine and along the large intestine results in haemorrhage and enteritis. Haemorrhagic diarrhoea and tenesmus are frequently observed in cases associated with E. bovis and E. zuernii. These species can cause significant disease and may result in mortality. E. alabamensis is moderately pathogenic and can lead to watery diarrhoea.5
Sheep
Lambs aged four weeks’ to six months’ are most susceptible to intestinal damage due to Eimeria infection.6 Multi-species infections are more common that single species in sheep and disease pathology may be severe, even with moderate infection doses. Common lesions associated with E. bakuensis are polyp formation in the small intestine due to local parasite replication. E. ovinoidalis can induce a severe and lethal enteritis soon after infection during the asexual stages of parasite replication.5
Diagnosis
Coccidiosis is often suspected based on farm history and presenting clinical signs. Patient age can be taken into consideration. Coccidiosis is rarely seen in animals less than three weeks old because the prepatent period of pathogenic species is generally between 15-21 days.2 Infection can occur soon after birth leading to disease in neonates from three weeks old if there is faecal and oocyst contamination of the calving or lambing pens.2
Subclinical forms are associated with depressed appetite and reduced feed conversion efficiency due to gut pathology leading to poor growth rates. Regular monitoring of feed intake and weight gain may be useful in detecting subclinical disease.
Clinical signs observed with clinical coccidiosis include diarrhoea (sometimes haemorrhagic), anorexia, abdominal pain, straining to defaecate, rectal prolapse, dehydration, depression and pale mucous membranes.2,6
Diarrhoea is the most common clinical sign and it may contain either digested or undigested blood depending on the site of intestinal lesions. The severity of disease varies from recovery without treatment to death. A nervous coccidiosis condition can occur in cases after heavy infection, mostly due to E. zuernii. Nervous signs such as muscle tremors, convulsions and nystagmus can occur. The nervous form of disease is uncommon but is associated with a high mortality rate.
Demonstration of causative pathogenic coccidia oocysts in faeces is key to diagnosis.1 In cattle, oocyst counts of greater than 500 per gram of faeces in combination with typical clinical signs are highly suggestive of coccidiosis.5 Low numbers of pathogenic coccidia oocysts justify further monitoring.5 It is important to note that oocysts are commonly shed by normal healthy animals in low levels. Non-quantitative tests are of limited value. Species identification is important as a high number of non-pathogenic species can infect ruminants.1
The clinical course of disease may be longer than the period of oocyst shedding. Clinical signs can occur during the prepatent period prior to the onset of oocyst excretion. Early samples may be negative so taking faecal samples from multiple animals in the group is indicated. Frequently clinical signs continue long after the patent period due to persistent pathology after parasite replication has ceased. Faecal analysis of animals during the chronic phase of disease may also provide a negative result.1,6 Therefore, key contributors to diagnosis include a knowledge of the farm history, animal signalment and clinical signs are useful along with accurate interpretation of faecal analysis.
In addition, oocyst excretion is higher during early patency and remains high for the first few days leaving the optimum time for sampling soon after the onset of diarrhoea rather than a week or more later.
Post-mortem examination can be useful to confirm the presence of coccidial species in a herd or flock. Intestinal haemorrhage and white/grey patches on the mucosa are indicative of coccidiosis. Histopathology can be confirmatory.6
Serological methods to detect antibodies in calves infected with E. bovis using ELISA and Western blot have been developed. However, they are not commercially available and not practical for calves that have received colostrum. Maternally derived antibodies may be detected that are not related to the current infection in samples taken from young calves. Furthermore, a possible cross reactivity between species may occur and antibodies may not relate to a primary response to infection or protective immunity. Serology is unlikely to be useful for individual animal diagnosis but may have a role in epidemiology and surveillance.1,5
Control
Disease prevention is key to control. Hygiene efforts intended to eliminate or reduce the build-up of faecal contamination have a limited success and effective control is not based on elimination of Eimeria from the farm.1,2,5 Strategies that minimise oocyst intake along with the administration of prophylactic coccidiostat drugs to infected animals during asexual reproductive stages of parasite development are most effective.1,2,5,6
Coccidiosis outbreaks are a herd-level problem and precipitated by stress. Therefore, minimising stressors such as mixing, re-grouping, dietary changes and overcrowding are key to reducing the risk of disease.5,6
The highest concentration of infective oocysts is found where the greatest amount of faecal matter builds up. Infection is via the faecal-oral route so management strategies that reduce faecal contamination of feed and water sources, along with good hygiene, will reduce the risk of disease occurring. Housing environment, hygiene, bedding, aeration, animal density and floor type should be evaluated.2
For highly pathogenic species, low-infective doses can cause disease and the prolific nature of the parasite rapidly leads to high re-contamination. Slatted floors and deep bedding can minimise oocyst intake from the ground. Oocyst sporulation can be reduced by maintaining a dry environment through good drainage and plenty of straw. Feeding from clean troughs that are elevated can reduce the risk of infection.1,2
If animals are fed using troughs outdoors, they should be moved regularly to avoid high levels of oocysts around feeding areas. A damp environment favours oocyst sporulation so water drinkers should be maintained in good repair to avoid overflow and pastures should be drained and wet areas fenced off.
Amine, cresol and chlorocresol-based disinfectants are effective against Eimeria oocysts. Correct application is key to their effectiveness. Cresol based disinfectants used at the recommended concentration inactivate oocysts after two hours of contact.1 Many products are inactivated by the presence of organic matter. Therefore, a prerequisite for effective disinfection is proper cleaning which can be difficult to achieve in many sheds. It has been doubted that improving shed hygiene is effective at controlling coccidiosis but can aid in reducing the infectious pressure.1,3 Exposure to sunlight can reduce oocyst survival.5,6
Pharmacological options
Prevention of coccidiosis is better than treatment because of both clinical and subclinical production losses. Furthermore, permanently damaged intestinal mucosa is unresponsive to treatment.6 Supportive therapy to reduce the severity of disease is required for individual clinical cases. While exposure to Eimeria oocysts is necessary for immunity to develop, this should be limited.6 When exposure to coccidiosis is known to occur, coccidiostat drugs are indicated to limit parasite replication and damage.1,5,6 There are three main approaches to dealing with coccidiosis using pharmacological agents: prophylactic, metaphylactic and therapeutic treatment.
Therapeutic treatment is of limited value as the life cycle of the parasite has already been completed and damage has occurred.1 Treatment may abrogate further development of oocysts. In an outbreak, clinical calves or lambs should be removed from the group to limit the spread to cohorts.1,6
The goal of prophylactic/metaphylactic treatment is to reduce the losses associated with infection during a known risk period. The aim is to disrupt reproduction of the parasite during the early stages of asexual replication to prevent multiplication and excretion of unsporulated oocysts in faeces.1 These drugs should preferably also act on the gamonts during the sexual reproduction stage as some animals in the group may have entered patency when treatment is administered.1 The main advantage of metaphylactic treatment is the establishment of species-specific partial immunity.5
There are two types of anticoccidial drugs: coccidiostatic and coccidiocidal. Coccidiostatic drugs inhibit parasite development and replication. However, as soon as administration ceases, parasite development can continue. These drugs should only be used prophylactically and be administered continuously while exposure is likely.
Coccidiocidal drugs kill the parasite. They mostly act on the asexual stage of parasite reproduction although some act on the sexual period also. These drugs are suited to pro-/metaphylactic treatment but can also be used to treat clinical cases during patency as they act on gamonts. Single administration to susceptible animals during risk periods is appropriate and re-treatment may be necessary if the period of risk is prolonged.1,2,5,6
Experience from the field and experimental studies show only limited efficacy of sulphonamides against mammalian coccidia. They also act against certain bacteria which may help to suppress secondary infections and may at least partially explain the benefit of sulphonamide treatment in coccidiosis outbreaks.1,2,5
Decoquinate is registered as anticoccidial in-feed medication in some countries and must be applied continuously over an extended period because only the initial phase of the coccidia life cycle (sporozoites and trophozoites) is controlled.1,2,5
Halofuginone is an anticoccidial that has been widely used in poultry production for control of Eimeria infections. In cattle the only indication for use of halofuginone is for the prevention and treatment of cryptosporidiosis of young calves.1
Benzene acetonitrile compounds (BAC) such as diclazuril and toltrazuril are known to act against various life cycle stages of Eimeria.1
The timing of a single administration of BAC approximately one week before the expected outbreak of coccidiosis may efficiently control coccidiosis. The optimal time to start treatment is after infection but before oocyst excretion and clinical disease thus allow immunity to establish but prevent full parasite development and intestinal damage.1,2,5
A single dose of diclazuril (1mg/kg BW) can control infections with E. bovis and/or E. zuernii when given before the onset of patency.1 BAC drugs can be used in an outbreak for the preventative treatment of infected animals. Coccidiosis may be supressed in infected cohorts approaching patency. Hence, treatment should include all calves in the group.1,2,5
In a 12-year study from 2006 to 2018 of ovine faecal samples submitted to Animal and Plant Health Association (APHA) laboratories in UK for coccidiosis analysis revealed an annual peak in positive results every year between April to June. Therefore, a single oral administration of BAC to lambs at four to six weeks of age at the time the coccidiosis can normally be expected on the farm is warranted. If there is high infection pressure, a second treatment may be indicated three weeks later.
Dairy calves kept in individual pens is associated with a low risk of infection although infection in the calving pen can occur. Moving to group pens is high risk for infection. Administering a dose to those susceptible animals Eight-15 days after moving is appropriate. This approach allows exposure to occur, immunity to develop and can be used for moving calves or lambs to contaminated pastures also.
Complete removal of oocysts from the environment is unlikely therefore a multimodal approach of good animal husbandry practices along with correct timing of BAC treatments to all animals in the group is a practical strategy to minimise the impact of coccidia infection among susceptible groups of calves and lambs on Irish farms.
-
Daugschies A, Najdrowski M. Eimeriosis in Cattle: Current Understanding. J. Vet. Med. B 52. 2005; 417-427
-
Jolley W, Bardsley K. Ruminant Coccidiosis. Vet Clin Food Anim 22. 2006; 613-621
-
Fox JE. Coccidiosis in cattle. Mod. Vet. Pract. 1985; 66:113-116
-
Grafner G. Further studies on the occurrence, epizootiology and control of Eimeria coccidiosis in cattle under the conditions of intensive stable housing. Mh. Vet. Med. 40. 1985; 41-44
-
Bangoura B, Bardsley K. Ruminant Coccidiosis. Vet Clin Food Anim 36. 2020; 187-203
-
Keeton S, Navarre C. Coccidiosis in Large and Small Ruminants. Vet Clin Food Anim 34. 2018; 201-208
-
Marquardt W. Effect of high temperature on sporulation of Eimeria zuernii. Exp Parasitol 1960; 10:58-65
-
Faber J, Kollmann D, Heise A, Bauer C et al. Eimeria infections in cows in the periparturient phase and their calves: oocyst excretion and levels of specific serum and colostrum antibodies. Vet. Parasitol. 2002: 106:1-17
-
Burger H. Eimeria infections in cattle. Berl. Mu¨nch. Veterinarian weekly 1983: 96, 350-357
1. Infection of ruminants by pathogenic Eimeria species is inevitable because of the following:
A. Oocysts are ubiquitous in the environment where animals are kept
B. Oocysts capable of surviving for long periods of time thus all calves become infected sooner rather than later
C. The risk of infection is increased where stocking density is high or faecal material accumulates coinciding with optimal environmental conditions for oocyst survival
D. The risk of succumbing to disease is increased when infected animals are exposed to high infectious pressure and management stressors at the same time
E. All of the above
2. Immunity to coccidiosis is:
A. Usually partial and strictly species-specific
B. Cross protective between species
C. Develops after little or no exposure
D. Always a reliable form of disease control
3. Coccidiosis is:
A. An individual animal problem that requires only clinical cases to be treated
B. A group problem requiring all animals in the group to be treated
C. Only an issue in young calves less than six months old
D. Rarely detected in lambs
4. Control of coccidiosis requires a multimodal approach including:
A. Prophylactic treatment using an anticoccidial product to the entire group prior to the period of risk
B. Improved hygiene
C. Appropriate stocking density
D. Adequate exposure to Eimeria species to allow immunity to develop
E. All of the above
Answers: 1E; 2A; 3B; 4E.















