Chronic kidney disease in cats – part 2
In the second part of this two-part series, Vanessa Bourne MVB CertAVP(SAM-F) PGCertVetEd FHEA MRCVS, Royal Veterinary College, continues her exploration of how to increase longevity, improve the client-practice bond and increase revenue by employing optimal monitoring and treatment options for chronic kidney disease in cats
Prognosis and management
Chronic kidney disease (CKD) is a very common finding in mature cats, potentially affecting over 30% of cats over 10 years of age. CKD is a generally slowly progressive disorder, caused by chronic tubulointerstitial nephritis and interstitial fibrosis in most cases. Patients can survive with reduced renal function for many years. Identification of complications and co-morbidities can allow for earlier interventions, improving quality of life and longevity. It therefore makes good business sense for clinicians to become familiar with the optimal monitoring and treatment options.
Management of CKD in feline patients
Management is focused on supportive care with interventions to reduce variables, which worsen disease (for example management of proteinuria) and those, which can affect morbidity (for example hypokalaemia). The goals are to improve quality of life and slow progression of disease. Administration of any nephrotoxic drugs should be discontinued where at all possible and careful consideration given to any medications in the future. As many drugs are metabolised or excreted by the kidney, reduction in renal function may affect concentrations of the drug and necessitate a dosage reduction or increase in treatment interval. Any pre- or post- renal diseases should be identified and treated. Radiography or ultrasound can be used to help identify conditions such as nephroliths or pyelonephritis. Urinalysis with urine culture can identify urinary tract infections or pyelonephritis.
Patients with CKD can develop acute-on-chronic disease. This refers to a variety of diseases where there is an acute worsening of renal function in a patient suffering from CKD. The most common cause of this seen in first opinion practice is the development of urinary tract infections. In many cases, the underlying cause can be determined and treated, reversing the decrease in the renal function or demonstrating an improvement from the acute deterioration.
Regular monitoring
There are a number of conditions and complications of CKD that can be identified through regular monitoring. These can either be treated or controlled in a first-opinion setting and include:
- Occurrence and progression of hypertension;
- Occurrence and progression of proteinuria;
- Occurrence and progression of hypokalaemia;
- Occurrence and progression of hyperphosphataemia;
- Occurrence and progression of CKD-MBD;
- Occurrence and progression of anaemia; and
- Occurrence of urinary tract infections or acute/chronic pyelonephritis.
Successful identification and management of these conditions slows the progression of CKD and improves the patient’s quality of life.
Maintenance of hydration
As CKD is associated with polyuria and polydipsia, affected cats may be at risk of developing dehydration, particularly in the later stages of disease. In some cases, patients may require hospitalisation and intravenous fluid therapy. This could apply to newly diagnosed patients or those who have developed acute-on-chronic disease. As well as fluid balance disorders, electrolyte and acid-base disturbances should be treated if identified. In the home environment, owners should be educated on how to maximise their pet’s water intake. This can take the form of a variety of containers placed around the house, usage of a water fountain, offering of wet food and so on. In some cases, it may be appropriate to consider the usage of subcutaneous fluid therapy or placement of a feeding tube. Subcutaneous fluids can be used for patients with advanced CKD, where maintaining hydration is difficult. A reasonable figure is 75-150ml administered every one to three days and can be given in the practice setting or by committed owners. Over supplementation can occur, potentially increasing blood pressure and causing the development of fluid overload. Therefore, it is important to establish a benefit for the individual animal and to educate the owner regarding possible adverse effects. The administration of subcutaneous fluids by owners in the home setting is common in the US but less common on this side of the Atlantic. There is no clear reason for this preference and as long as owners are well informed on potential complications that may develop, no reason why owners cannot be educated as to how to perform this simple treatment themselves. Information including demonstration videos can be found at https://icatcare.org/advice/how-to-give-subcutaneous-fluids-to-your-cat/
In some cases, placement of an oesophagostomy feeding tube can provide access for fluids, nutrition and medications. Some cats will tolerate feeding tubes very well and these can be left in situ for a prolonged time, as long as the owner is able to manage the tube at home. This can raise ethical concerns, so careful case selection is important.
Dietary management for IRIS CKD stages 2-4
Renal diets are restricted in both protein and phosphorus. They have an increased calorie content, are alkalinising, restricted in sodium and supplemented with potassium, B vitamins, antioxidants and omega-3 fatty acids. Currently, there is good evidence to demonstrate that patients in IRIS stage 2-4 CKD have increased longevity when they consume a renal diet. Renal diets have also been shown to reduce uraemia. There is no current evidence to suggest a benefit in patients with IRIS stage 1 CKD. Moderate protein restriction is currently recommended in CKD as excess restriction may result in a reduction in muscle mass. Hyperphosphataemia is known to be associated with a progression of CKD. As renal diets are restricted in phosphate, feeding this alone can be sufficient to reduce the phosphate concentrations into the target ranges per IRIS stage. Where an excess in phosphate is identified, the first step is to transition the patient onto a renal diet and then re-measure the phosphate level approximately four weeks after the change. If phosphate remains elevated, or the patient is already on a renal diet, phosphate binders should be introduced.
Several binders are available. If the patient does not tolerate one, another should be tried as palatability will vary between products. Care should be taken when using calcium containing phosphate binders, as this could promote hypercalcaemia. Therefore, serial monitoring of ionised calcium is recommended in these cases.
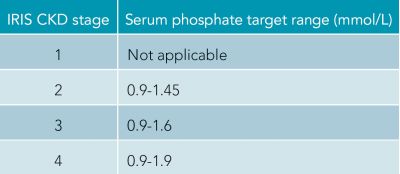
Figure 1: IRIS recommended target serum phosphate concentrations.
Renal diets can be less palatable, so a gradual transition is required. It is not usually suitable to try and implement a renal diet while a patient is being hospitalised for treatment of CKD as this can create long-term food aversion. Placing medications in food is similarly discouraged. Cats with CKD can have excessive excretion of potassium into the urine, leading to hypokalaemia. Hypokalaemia can cause lethargy, inappetence, constipation and muscle weakness. It may also contribute to the development of acidosis. Renal diets are supplemented with potassium. Hyperkalaemia can also occur in advanced CKD. It is recommended to serially monitor serum potassium levels. Supplementation with potassium gluconate is generally recommended if serum potassium concentrations are <3.5 mmol/L, with the dosage adjusted depending on response. A typical starting dose is 1-4mmol of potassium per cat q12h.

Figure 1: With monitoring and necessary treatment implementation, survival of cats with CKD can be prolonged and quality of life improved.
Management of hypertension in patients with CKD
Hypertension is usually considered to be present with a systolic blood pressure (SBP) persistently in excess of 160-180mmHg, but can also be considered present if there is clear indication of target organ damage (TOD). Affected organs include the kidneys, brain, eyes and heart. The kidneys are able to autoregulate, controlling the blood pressure within the organ to an extent and maintaining renal blood flow and GFR over a range of SBP around 80-160mmHg. Outside of these limits or where CKD is present, elevated pressure to the glomerular capillaries results in glomerular hypertension and the potential for glomerulosclerosis.
Treatment aims include a reduction of SBP to 120-150mmHg (<160 mmHg may be acceptable in some cases). In most cases of cats with hypertension and CKD, a gradual reduction in blood pressure is indicated, avoiding hypotension. Detection of elevated SBP on one occasion is not enough to initiate treatment unless there is clear indication of TOD on physical examination. SBP should be re-measured in one to two weeks in patients where a reading of >160mmHg has been obtained.
The treatment of choice is amlodipine, a calcium channel blocker (CCB), with a starting dose of 0.125-0.25mg/kg orally once daily. Telmisartan, an angiotensin receptor blocker (ARB), has recently been licensed for usage as an anti-hypertensive medication at 2mg/kg orally once daily. It is more commonly used in the treatment of proteinuria, at 1mg/kg orally once daily. Angiotensin-converting enzyme inhibitors (ACEI) have a limited effect in the reduction of blood pressure, typically reducing SBP by 10-15mmHg. They are however useful in the treatment of proteinuria.
If the desirable reduction in SBP has not been achieved, the dosage or amlodipine can be doubled (0.25-0.5 mg/kg once daily). Telmisartan dosages cannot be increased. A combination of amlodipine and telmisartan can be considered if neither drug alone does not adequately control blood pressure. CCB or renin-angiotensin-aldosterone system inhibitors (ARB or ACEI) should not be introduced to patients that are dehydrated as a precipitous drop of GFR can occur. In patients with advanced CKD and therefore reduced GFR, care should also be taken.
Treatment is generally life long, with treatment adjustments possible as disease progresses. Monitoring of SBP should be performed at least every three months on stable patients, with more frequent reassessments of unstable patients. SBP <120mmHg should be avoided. Clinical signs of hypotension include weakness and tachycardia. A small and persistent increase in creatinine (<45µmol/L) can occur with reduction in blood pressure. More marked increases can suggest an adverse drug effect with progressive increases indicating a progression of kidney disease.
Management of proteinuria in patients with CKD
Proteinuria should be treated when there are persistent UPC measurements are >0.4. If proteinuria is detected on a single sample, this should be repeated after two to three weeks to ensure persistence and assess magnitude. Some clinicians recommend treatment of proteinuria in the range 0.2-0.4, because the association between progression of CKD and proteinuria includes the borderline category. However, there is no current evidence that intervention with anti-proteinuric drugs slows progression.
In cats, inhibition of the renin-angiotensin-aldosterone system (RAAS) has been shown to reduce the magnitude of proteinuria. Benazepril, an angiotensin converting enzyme inhibitor (ACEI), is frequently used at a dosage of 0.25-0.5mg/kg PO q24h. It undergoes hepatic metabolism so may be preferable to those undergoing renal metabolism. Alternatively, an angiotensin receptor blocker (ARB), telmisartan can be used at a dosage of 1mg/kg PO q24h. Hypertension can cause proteinuria; therefore it is essential to measure SBP in patients. Where hypertension and proteinuria are identified, it is recommended to treat the hypertension first and reassess the magnitude of proteinuria, as antihypertensive treatment alone may successfully reduce proteinuria to target range.
Administration of RAAS inhibitors can reduce GFR and cause a concurrent increase in creatinine. Usage of RAAS inhibitors should only be used in stable patients that are hydrated, otherwise a dangerous drop in GFR can occur. This can also be more likely to occur in cats with more advanced CKD in Stages 3 and 4. Therefore caution should be used with introduction of these medications. Goals of treatment include a decrease in UPC while maintaining a stable creatinine.
Increasing creatinine values or an increase in UPC indicate that disease is progressing. Treatment is generally lifelong unless the underlying disease resolves (unlikely in most feline cases).
Management of anaemia in patients with CKD
Anaemia can occur in CKD, due to a reduction in erythropoietin produced by the kidney. Blood loss and shortened red blood cell survival can also occur in patients with CKD. Anaemia has been associated with progression of CKD. It can be non- or poorly- regenerative in nature.
Some patients may benefit from the usage of erythrocyte stimulating agents (ESA). There has been a trend to use darbepoetin, as this may be associated with a decreased risk of adverse effects. ESA usage should be considered for patients with a PCV persistently below 20% or where there are clinical signs associated with the anaemia. Concurrent iron supplementation is recommended. The dosage of darbepoetin is 1µg/kg SC once weekly until the PCV is above or equal to 25%. The dosage interval can then be increased to every two to three weeks or a lower dose can be given weekly. Some animals will not respond to therapy and reasons for this include concurrent illnesses, infections, inflammation, gastrointestinal bleeding, iron deficiency or the development of pure red cell aplasia from production or anti-darbapoetin antibodies. The development of hypertension has also been reported.
Management of inappetence, nausea or vomiting in patients with CKD
Cats with advancing CKD can suffer from periods of inappetence, nausea or vomiting due to uraemic toxins triggering the chemoreceptor trigger zone in the brain. Inappetence can also occur if the patient is suffering from other complications of CKD, for example anaemia, hypokalaemia, dehydration or metabolic acidosis.
Maropitant, a neurokinin-1 (NK-1) receptor antagonist, is the most commonly used anti-emetic. It may not have an effect on nausea. The dosage is 1mg/kg q24h via subcutaneous or intravenous injection or 2mg/kg q24h orally. Ondansetron, a serotonin receptor (5-HT3) antagonist, may be more successful at treating nausea, at a dosage of 0.5-1mg/kg q24h via subcutaneous injection or orally. Mirtazapine, a tetracyclic antidepressant and alpha-2 antagonist, is commonly used as an appetite stimulant and can help to reduce vomiting. It can also have adverse effects including behavioural changes so should be used with caution. The dosage in cats with CKD is 0.5mg/kg or 1.88mg/cat q48h orally.
There are some reports of gastroprotectants, such as histamine receptor (H2) blockers or proton pump inhibitors (PPIs) improving inappetence in cats with CKD, however efficacy remains unproven. Where clinicians are prioritising treatments where financial restrictions exist or there are issues with administration of medications, it would be preferable to first select proven medications to control nausea.
There are reports of successful long-term placement of oesophagostomy feeding tubes in the management of CKD. These allow administration of fluids, food and medications. There are some ethical concerns raised around the placement of enteral feeding tubes and careful consideration should be made in each case of whether this is an appropriate course of action.
Management of urinary tract infections in patients with CKD
Bacterial urinary tract infections (UTIs) occur at a rate of around 15-30% of cats with CKD, with older female cats being predisposed. Many infections are subclinical, meaning that the patient has no clinical signs of lower urinary tract disease. In these cases pyuria, bacteruria or haematuria may be noted on urinalysis. Nonspecific clinical signs of lower urinary tract disease include pollakiuria, dysuria, stranguria, haematuria and inappropriate urination.
Pyelonephritis can result from ascending infections and can be acute or chronic in nature. Acute disease is likely to have signs of systemic illness including pyrexia, abdominal pain, possible enlarged kidneys and changes on blood profiles (azotaemia, white blood cell changes). Chronic pyelonephritis has a more insidious onset and may result in a slowly progressive azotaemia.
Escherichia coli is the most common cause of UTIs, followed by gram-positive cocci and then various others including Proteus, Pseudomonas, and Klebsiella species also being found. The significance of subclinical bacteriuria and treatment is currently controversial. Some clinicians recommend treatment due to the risk of ascending infection to the kidneys as a result of a reduction in host defence mechanisms. Recurrence of UTIs post treatment or the development of antibiotic resistance in the patient is possible.
- Samples for urine culture should be obtained by cystocentesis and submitted in plain tubes, not boric acid.
Treatment of UTIs in cats with CKD is currently recommended where there is a positive urine culture and where there are signs of lower urinary tract disease (for example dysuria, pollakuria) and/or systemic signs are present (for example pyrexia, white cell count changes, renal pain) and/or pyuria is present (>5WBCs per hpf) and/or there is an unexpected deterioration in renal function.
Antibiotic selection should be based on culture results and whether the antibiotic has an excellent urine penetration. First line antimicrobials for uncomplicated UTIs include cephalexin (15-30mg/kg q12h PO) or amoxicillin (11-15mg/kg q8h PO). Potentiated amoxicillin (12.5mg/kg q12h PO) is reserved for complicated UTIs, as are fluoroquinolones and extended-release cephalexin (cefovecin).
A simple uncomplicated UTI refers to infections occurring in otherwise healthy animals. Infections in patients with CKD can be classified as complicated, as there is an underlying concurrent disease present. In these cases, treatment is normally recommended for two to four weeks but it may be necessary to treat for up to six weeks. Success of treatment is normally determined by repeat culture seven days after the antibiotics course has finished.
Pyelonephritis would also be classified as a complicated UTI, as it involves the upper urinary tract. A positive result is helpful for diagnosis; however, a negative result does not exclude pyelonephritis. Diagnosis may be presumptive based on urine culture results, diagnostic imaging findings and an improvement in azotaemia in response to treatment.
Acute pyelonephritis requires hospitalisation for intravenous fluids and antibiotics (plus other supportive measures). Infections should be treated for a minimum of six to eight weeks. While both acute and chronic pyelonephritis would be classed as complicated UTIs, chronic disease can usually be treated on an outpatient basis. Fluoroquinolones are a good first line choice (for example marbofloxacin 2mg/kg q24h PO or IV).
Identification of abnormalities in plasma phosphate, UPC and red blood cells with subsequent successful treatment and continued monitoring can therefore increase longevity in patients with CKD. FGF-23 measurement in practice is not currently available, but may become more accessible in the future. Feeding a renal diet has been demonstrated in multiple studies to help moderate plasma phosphate and levels of azotaemia, reducing clinical signs of uraemia and prolonging longevity. One study demonstrated a median survival for cats receiving a therapeutic renal diet was almost three times longer than cats receiving a maintenance diet.
Summary
There are a number of interventions and monitoring techniques available for our geriatric cats with CKD.
Using these improves the longevity and welfare of our patients, as well as increasing income and solidifying the client-practice bond.
Prognosis for cats with CKD
-
The prognosis for cats with CKD depends on the severity of disease, which can be related to their IRIS staging. IRIS stage of CKD based on serum creatinine at the time of diagnosis is strongly predictive of survival in cats with naturally occurring CKD. Cats diagnosed in the early stages of disease have been shown in multiple studies to survive for much longer than animals initially diagnosed with CKD at a later stage of disease. There are several additional parameters associated with a worse prognosis. These are:
-
Documented progression of CKD (reflected by measured increases in creatinine)
-
The level of hyperphosphataemia
-
The level of proteinuria
-
Anaemia
-
Elevated FGF-23
1. What parameters have been associated with a poorer prognosis in patients with CKD?
A. Hyperphosphataemia, proteinuria, anaemia
B. Hyperphosphataemia, hypokalaemia, proteinuria
C. Hypokalaemia, hypertension, anaemia
D. Proteinuria, anaemia, hypertension
2. What medications are most useful in the management of hypertension?
A. Amlodipine
B. Telmisartan
C. Benazepril
D. A or B
E. A or B or C
3. What medications are most useful in the management of proteinuria?
A. Amlodipine
B. Telmisartan
C. Benazepril
D. B or C
E. A or B or C
4. The long-term goal of hypertension management is to maintain blood pressure within the following range:
A. 120-150mmHg
B. 90-150mmHg
C. 120-180mmHg
D. 150-200mmHg
5. Bacterial UTI occur in which percentage of cats with CKD?
A. 15-30%
B. <2%
C. 50%
Answers: 1A; 2D; 3D; 4A; 5A















