Large animal - February 2019
The multifaceted role of calcium in milk fever
John Lawlor, ruminant technical manager, Anchor Life Science Ltd, examines the multifaceted role that calcium plays in milk fever and its associated metabolic issues. A greater understanding of these factors may be one way to capitalise on the post-quota opportunities on Irish dairy farms, he writes
Dairy farming in Ireland has seen many changes over the past 10 years, especially since the dawning of the post-quota era. Milk fever continues to be a major challenge and cost, not only in terms of cow health and lost productivity, but also in terms of scarce labour.
Over the past decade, Irish dairy farmers have embraced new ideas and opportunities to develop their businesses such as: post-quota expansion; more compact calving patterns; and a greater focus on milk from grass. However, a compact calving season when combined with more cows and a lack of skilled labour, means that the calving season is the most stressful time of year on most dairy farms. The calving season also coincides with the period when farms are most exposed to the second most costly disease on the dairy farm, milk fever. It is well established that calcium (Ca) plays a major role when it comes to milk fever and that cows with milk fever are more prone to other costly metabolic issues (Curtis, 1983). Recent research has shown that sub-clinical milk fever has a major unseen cost on farm and is responsible for 80% of the annual cost of milk fever. Reducing the risk of milk fever, clinical and sub-clinical, must be a key priority on Irish dairy farms, due to the large impact it has on herd health, production and profitability.
Milk fever prevalence and economic cost
Over the years, numerous studies have tried to quantify the incidence level and cost of clinical milk fever (blood Ca of <5.5mg/dl). DeGaris and Lean (2009) found that the average incidence level in 10 European studies was 6.2% (range 0-10). Numerous other studies have reported clinical milk fever incidence to be in the order of 5-7% (NAHMS 2002; Goff 2008; Mulligan and Doherty 2008). In 2011, Oetzel estimated the cost of a single case of clinical milk fever at $300. Until relatively recently, our understanding of the incidence level and economic cost of sub-clinical milk fever (blood Ca 5.5-8.0mg/dl) has not been very clear. Work by Reinhardt et al (2011) put the scale of the problem into focus. In this study, Reinhardt found that approximately 50% of second lactation and older cows were in the sub-clinical range and, perhaps surprisingly, that 25% of first lactation heifers were also in the sub-clinical range. A more recent French study (Millemann et al, 2016) found similar results in terms of the level of subclinical milk fever in multiparous cows at 57% and again an incidence level in first lactation heifers of 16%. Oetzel (2011) also found that a case of sub-clinical milk fever costs $125.
Given that many more cows are at risk of sub-clinical milk fever than clinical, these studies demonstrated that 80% of the annual cost of milk fever is coming from the sub-clinical milk fever, not clinical cases.
Calcium in the cow
Ca is the most abundant mineral in the cow, however 99% of it is to be found in bones and, as such, is not readily available to the cow. The cow’s homeostatic mechanism to maintain normal blood calcium operates efficiently most of the time with normal blood Ca levels held within a narrow range (8.5-10mg/dL). The Ca in the cow’s bones is in a state of continuous exchange with the extracellular fluids. There is about 10-11g of Ca available in the extracellular pool, where it plays an important role in muscle contraction and nerve function. A cow can lose 9-13% of her skeletal Ca during the first month postpartum (Ellenberger et al, 1932).
Of the extracellular Ca pool, there is ~2.5-3.5g of Ca which is held within the plasma Ca pool, and which is found as protein-bound Ca, Ca bound in complexes with anions and ionised Ca. Correct muscle and nerve functions rely on the maintenance of an adequate and consistent level of ionised Ca. The plasma pool may need to replenish itself between eight and 10 times per day in early lactation, to support milk production and maintenance of the cow. The remaining calcium pool is the intracellular Ca pool. Intracellular Ca plays a key role in immune function as its signalling is a key early feature in immune cell activation.
Milk fever causes
Milk fever or hypocalcaemia is a common disease of calving or freshly calved cows. The sudden increase in demand for Ca at the onset of lactation, presents a major challenge to a cow’s homeostatic control mechanism. The onset of lactation increases the demand for Ca by several fold. The cow’s normal reserve pool of Ca is about 2.5-3.5g and cows can only afford to lose approximately 50% of this pool before a hypocalcaemia crisis is initiated. With a single litre of colostrum requiring 2.3g of Ca, it is easy to see how a cow can quickly become Ca deficient. It is not so much the sudden demand for Ca that causes milk fever but more so the fact that the cow’s homeostatic system can take 24-48 hours post calving to become fully functional. It is this time-lag that causes the cow to drain Ca from her plasma pool and as this reserve decreases so too does the cow’s blood Ca status, possibly bringing about a case of sub-clinical or clinical milk fever.
Homeostatic mechanism
Ca supply during periods of hypocalcaemia is controlled by two key hormones: parathyroid hormone (PTH) and vitamin D3. PTH is also produced by the thyroid gland and its secretion is stimulated by low blood Ca concentrations. Vitamin D3 itself is quite inactive and must undergo two chemical processes to become active. In the liver it is converted to 25-hydroxy-Vit D3 and undergoes a further chemical conversion in the kidney to 1-25-dihydroxy-Vit D3. The second conversion is reliant on PTH, while the first is reliant on magnesium and may be compromised under conditions of hypomagnesia. As colostrum/milk production increases rapidly, so too does the drain on the cow’s Ca reserve pool, causing the concentration of blood Ca to fall. This then triggers the secretion of PTH by the parathyroid gland and consequently the conversion of 25-hydroxy-Vit D3 to 1-25-dihydroxy-Vit D3.
Bone Ca mobilisation is now stimulated by both PTH and Vit D3 but can take up to 48 hours to respond fully. Absorption of Ca from the intestines is solely under the control of Vit D3 and can take 24 hours of Vit D3 stimulation before calcium absorption increases significantly. It is this 24-48-hour time lag that causes blood Ca levels to drop.
Other important minerals
Magnesium
When it comes to milk fever, the role of magnesium (Mg) cannot be understated. Mg is critical for the release of PTH and in the synthesis of 1-25-dihydroxy-Vit D3. Mg stored in the soft tissues and bone of the cow is unavailable to the cow and so she requires a continuous dietary supply to meet her requirements.
Moderate hypomagnesemia reduces the ability of the cow’s homeostatic control mechanism to function properly and hypocalcaemia occurs. Hypomagnesemia can affect Ca metabolism by:
a) reducing PTH secretion in response to hypocalcaemia; and
b) reducing tissue sensitivity to PTH (Rude, 1978).
Hypomagnesemia can occur in cows either fed an inadequate amount of Mg in the diet or if the diet contains high levels of some other factor that prevents adequate absorption of magnesium, such as potassium.
Potassium
Potassium (K) binds up Mg in the rumen, which can subsequently inhibit the availability of calcium to the cow. High levels of dietary K are common in Irish grass silages particularly second and third cut silage, which may have received high levels of manure, and which is typically fed to dry cows and youngstock. The recommended dietary K level in the diet of a dry cow is ≤1.8%DM, yet Rogers and Murphy (2000) report that the mean level in Irish grass silages at 2.35% with a range from 0.63%-5.59%. They also report that 11% of Irish grass silages have a K level exceeding >3.1%. Apart from binding Mg, K also has an alkalising effect and can contribute to metabolic alkalosis, a further risk factor for milk fever.
Phosphorous
High phosphorous (P) concentrations have been shown to result in increased milk fever risk. Blood P levels, above 2mmol/L, can inhibit the enzyme converting 25-hydroxy-Vit D3 to 1-25-dihydroxy-Vit D3. This can compromise the ability of the cow to produce the hormone necessary for the activation of intestinal Ca transport, which will impair the Ca homeostasis of the cow.
Other risk factors
Metabolic alkalosis
Pre-calving diets supply greater levels of cations (K, Sodium [Na], Ca and Mg) than anions (chlorine [Cl] and sulphate [SO4] and phosphate [PO4]) cause a greater number of positively charged cations than negatively charged anions to enter the blood, which increases blood pH, resulting in a state of metabolic alkalosis. Metabolic alkalosis blunts the response of the cow to PTH (Gaynor 1989; Goff et al, 1991; Phillippo et al, 1994), which increases the risk of milk fever and subclinical hypocalcaemia (Craigie and Stoll, 1947).
Age
Older cows have a decreased capacity to mobilise Ca from their bones (Van Mosel et al, 1993) and possibly a decreased number of 1-25-dihydroxy-Vit D3 receptors in the small intestine (Horst et al, 2003). Lean (2006) found that the risk of milk fever increases by 9% per lactation.
Condition score
Over-conditioned cows (body condition score >3.5 on a 1-5 scale) are at an increased risk of hypocalcaemia (Heuer et al, 1999).
Other common risk factors include: channel island breeds (Harris, 1981); high-yielding cows; lame cows; and cows with a previous history of milk fever (Erb et al, 1985).
Multifaceted role of Ca
Ca plays a critical role to the transitioning cow and its effect is broad based, as Ca plays an important role in a number of key areas for the cow in transition, from muscle and nerve function, to the physical barriers of the innate immune response and also the phagocytic response.
Muscle function
As calcium is required for proper muscle function, cows with low blood calcium levels lose muscle tone and this has a number of negative effects on key muscular functions of the cow.
Mobility
One of the classic and early indications of milk fever is a cow with an unsteady and staggered walk. Hypocalcaemic cows may be less mobile, leading to lower feed intakes with possible negative effects on energy status. Hypocalcaemic cows can be more prone to slipping, falling and injury leading to higher culling rates.
Impaired rumen activity
Huber et al (1981) found that rumen contractions ceased well before the onset of signs of clinical milk fever in artificially induced hypocalcaemia. Such stasis may be an important factor in the development of hypocalcaemia as even the temporary alimentary stasis can induce acute hypocalcaemia through reduced intestinal absorption of Ca (Moodie and Robertson, 1962). A further study (Jorgensen et al, 1998) showed the negative effects of hypocalcaemia on smooth muscle contractility in cows artificially induced with progressively severe hypocalcaemia. The study demonstrated a significant reduction of contractility even with mild subclinical hypocalcaemia (0.78mmol/L ionised Ca). Reduced feed intakes can lead to an impaired energy status and a higher risk of displaced abomasum.
Innate immunity – physical barriers
Increased mastitis risk
The ability of the teat sphincter muscle to close the teat canal properly post milking is reduced in hypocalcaemic cows which may lead to a higher risk of mastitis. (Kimura et al, 2006). Curtis (1983) reported that cows with hypocalcaemia were eight times more likely to suffer from a case of mastitis.
Tight junction permeability
Ma et al (2000) demonstrated the role extracellular calcium levels play in the permeability of the intestinal tight junction, a key physical barrier to infection. The study was designed to study the role that extracellular Ca++ plays in the modulation of the intestinal epithelial monolayer tight junction barrier. The study demonstrated that extracellular Ca++ is crucial for the maintenance of intestinal epithelial tight junction barrier function.
Innate immunity – phagocytic response
Parturition places a lot of stress on dairy cows and is linked with significant immune suppression and increased susceptibility to infectious disease. The intracellular reserve of Ca is responsible for signalling within the cell and initiation of the immune response to bacterial challenges. Kimura et al, (2006) demonstrated that hypocalcaemia directly reduces immune cell response to an activating stimulus. This study suggested that intracellular Ca stores were being utilised to help sustain extracellular Ca or that when the cow is struggling to maintain Ca homeostasis, the ability to keep endoplasmic reticulum Ca stores fully stocked is compromised. This reduction in intracellular calcium stores in immune cells could blunt intracellular calcium release following an activating stimulus, contributing to the immune suppression seen in hypocalcaemic cows.
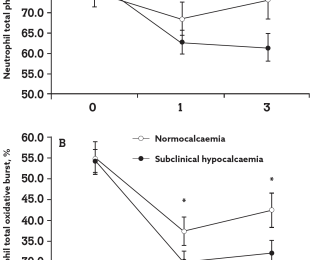
A further study by Martinez et al, (2012) demonstrated the link between immune function and calcium status in freshly calved cows. This study found that the neutrophil concentration in blood and the percentage of neutrophils undergoing phagocytosis and oxidative bursts were all reduced in cows with subclinical hypocalcaemia (blood Ca of <8.59mg/dl) compared to normo-calcaemic cows.
The study reported that, not only did the sub-clinical hypocalcaemic group have neutrophils with reduced in-vitro phagocytic and killing activities (graph B) but they also had a smaller leukocyte population in the blood (graph A) because of a sharp decline in neutrophils compared to normocalcaemic cows.
Martinez et al (2012) also found that sub-clinical hypocalcaemic cows had a higher risk of metritis and puerperal metritis compared to normo-calcaemic cows in both study sub populations. Cows were divided at calving based on their calving experience and divided into two groups: low-risk group – cows with a normal calving event; and high-risk group – cows who calved with dystocia, still birth, retained placenta, twins or a combination of issues. The study found that the risk of metritis decreased by 22% for every 1mg/dl increase in blood calcium level recorded over the first three days after calving.
Practical prevention strategies
There are numerous practical proactive preventative strategies available that can help to reduce the risk of milk fever at calving:
- Low calcium pre-calving diets – restricted dietary Ca will prime the homeostatic response. A target level of less than 30g Ca/day (20g of absorbable Ca) is required but can be difficult to achieve with background levels of Ca in forage;
- Anionic salts – addition of anionic salts in the last three weeks pre-calving will prompt a metabolic acidosis in order to counteract cationic elements in the diet and will prime the homeostatic response. This requires two separate dry-cow groups to administer and needs to be adequately mixed into the diet, can be hard to achieve without a diet mixer;
- Oral Ca supplementation – Ca boluses given at and 12 hours after calving have become a popular way to support blood Ca levels in the cow, during the period of highest demand. The use of oral Ca supplements allows for a targeted approach of high-risk cow groups within the herd. Liquid, gel and paste oral Ca supplements are also available.
Conclusion
Ca plays a central role in milk fever and having a clear management protocol in place can go a long way to keeping milk fever and its associated metabolic issues at bay. Whilst Ca and its management are key, it is certainly not the only risk factor that needs to be addressed. Energy status, body condition score, and adequate feed barrier space, are just some of the many management areas that require adequate attention and action in order to improve the success of the transition period. Reducing the risk of milk fever, clinical and sub-clinical, must be a key priority on Irish dairy farms, due to the large impact it has on herd health, production and profitability. Optimising Ca status at calving must be a part of any herd management protocol during calving season.
- DeGaris, P.J., Lean, (2009). Milk fever in dairy cows: A review of pathophysiology and control principles. The Veterinary Journal 176, 58-69.
- NAHMS (National Animal Health and Monitoring System), (2002). Dairy 2002 Report Part I: Reference of Dairy Health and Management in the United States. Natl. Anim. Health Monit. Syst., US Dept. Agric., Anim. Plant Health Insp. Serv., Vet. Serv., Cent. Epidemiol. Anim. Health, Fort Collins, CO
Goff, J.P., (2008). The monitoring, prevention, and treatment of milk fever and subclinical hypocalcemia in dairy cows. The Veterinary Journal 176, 50-57. - Mulligan FJ and Doherty ML. (2008). Production diseases of the transition cow. The Veterinary Journal, 176, 3-9
Oetzel, G. R., (2011). An update on hypocalcemia in dairy herds. School of Veterinary Medicine, University of Madison, Wisconsin: 80-85. - Reinhardt T A, Lippolis JD, McCluskey BJ, Goff JP & Horst, RL. (2011). Prevalence of subclinical hypocalcaemia in dairy herds. The Veterinary Journal 188, 122-124.
- Millemann Y, Gillet M, Kirsch P, Besnier P. (2016). Subclinical hypocalcaemia in French dairy cows. Presentation at World Buiatrics Congress, Dublin 2016.
- Ellenberger HB, Newlander JA, Jones CH. (1932). Calcium and phosphorus requirements of dairy cows. II. Weekly balances through lactation and gestation periods. Vermont Agricultural Experiment Station, Bulletin 342, June 1932.
- Goff, JP, (2006). Macromineral physiology and application to the feeding of the dairy cow for prevention of milk fever and other periparturient mineral disorders. Animal Feed Science and Technology 126, 237-257.
Rude, RK, Oldham, SB, Sharp Jr CF, Singer FR. (1978). Parathyroid hormone secretion in magnesium deficiency. J. Clin. Endocrinol. Metab. 47, 800-806. - Rogers PAM and Murphy R. (2000). Levels of Dry Matter, Major Elements (calcium, magnesium, nitrogen, phosphorus, potassium, sodium and sulphur) and Trace Elements (cobalt, copper, iodine, manganese, molybdenum, selenium and zinc) in Irish Grass, Silage and Hay. Available at: http://homepage.eircom.net/~progers/0forage.htm
- Gaynor PJ, Mueller FJ, Miller JK, Ramsey N, Goff JP, Horst RL. (1989). Parturient hypocalcemia in jersey cows fed alfalfa haylage based diets with different cation to anion ratios. Journal of Dairy Science 72, 2525-2531.
Goff JP, Horst RL, Mueller, FJ, Miller JK, Kiess GA, Dowlen HH. (1991). Addition of chloride to a pre-partal diet high in cations increases 1,25-dihydroxyvitamin D response to hypocalcemia preventing milk fever. Journal of Dairy Science 74, 3863-3871. - Phillippo M, Reid GW, Nevison IM. (1994). Parturient hypocalcaemia in dairy cows: effects of dietary acidity on plasma minerals and calciotrophic hormones. Research in Veterinary Science 56, 303-309.
Craige AH, Stoll IV. (1947). Milk fever (parturient paresis) as a manifestation of alkalosis. American Journal of Veterinary Research 8, 168. - van Mosel M, Van’t Klooster AT, van Mosel F, Kuilen JVD. (1993). Effects of reducing dietary [(Na++K+) – (Cl- + SO=4) on the rate of calcium mobilisation by dairy cows at parturition. Research in Veterinary Science 54, 1-9.
Horst RL, Goff JP, McCluskey BJ. (2003). Prevalence of subclinical hypocalcemia in US dairy operations. Journal of Dairy Science 86 (Suppl. 1), 247. - Lean IJ, DeGaris PJ, McNeil DM, Block E. (2006). Hypocalcemia in dairy cows: meta analysis and dietary cation anion difference theory revisited. Journal of Dairy Science 89, 669-684.
Heuer C, Schukken YH, Dobbelaar P. (1999). Postpartum body condition score and results from the first test day milk as predictors of disease, fertility, yield and culling in commercial dairy herds. Journal of Dairy Science 82, 295-304. - Harris DJ. (1981). Factors predisposing to parturient paresis. Australian Veterinary Journal 57, 357-361.
Erb HN, Smith RD, Oltenacu PA, Guard CL, Hillman RB, Powers PA, Smith MC, White ME. (1985). Path model of reproductive disorders and performance, milk fever, mastitis, milk yield and culling in Holstein cows. Journal of Dairy Science 68, 3337-3349. - Huber TL, Wilson RC, Stattleman AJ, Goetsch DD. (1981). Effect of hypocalcaemia on motility of the ruminant stomach. American Journal of Veterinary Research 42, 1488-1490.
Moodie EW, Robertson A. (1962). Some aspects of calcium metabolism in the dairy cow. Research in Veterinary Science 3, 470-484. - Jorgenson RJ, Nyengaard NR, Hara S, Enemark JM and Andersen PH. (1998), Rumen motility during induced hyper- and hypocalcaemia. Acta. Vet. Scand. 39:331-338.
- Kimura K, Reinhardt TA, Goff, JP, (2006). Parturition and hypocalcemia blunts calcium signals in immune cells of dairy cattle. Journal of Dairy Science 89, 2588-2595.
- Curtis CR, Erb HN, Sniffen CJ, Smith RD, Powers PA, Smith MC, White ME, Hillman RB and Pearson EJ. (1983). Association of parturient hypocalcemia with eight periparturient disorders in Holstein cows. J. Am. Vet. Med. Assoc. 183, 559-561.
- Ma TY, Tran D, Hoa N, Nguyen D, Merryfield M and Tarnawski A. (2000). Mechanism of Extracellular Calcium Regulation of Intestinal Epithelial Tight Junction Permeability: Role of Cytoskeletal Involvement. Microscopy Research and Technique 51, 156-168.
- Martinez N, Risco CA, Lima FS, Bisinotto RS, Greco LF, Ribeiro ES, Maunsell F, Galvão K and Santos JEP. (2012). Evaluation of peripartal calcium status, energetic profile, and neutrophil function in dairy cows at low or high risk of developing uterine disease. J. Dairy Science 95, 7158-7172.