BTM and national surveillance of jd
Liam Doyle MVB MSc PhD, AHI programme manager for Johne’s disease, discusses bulk tank milk testing (BTM) and national Johne’s Disease surveillance
Herds affected with Johne’s disease (JD) can have reduced milk production and higher rates of mastitis, infertility and lameness, even before the severe signs of disease in individual animals become apparent. Individual cattle with infection can show severe weight loss and diarrhoea, leading to death. Also in terms of on-farm methane production, JD is listed as one of the key diseases considered to negatively impact methane emissions from ruminants.1,2
Cattle are usually infected as calves by ingesting (swallowing) Mycobacterium avium paratuberculosis (MAP) bacteria from colostrum or milk or from their environment. The bacteria are shed in dung from infected animals, especially adult cattle, to contaminate the local environment including bedding and udders and teats.
The bacteria may also be shed directly in colostrum and milk. Calves may also become infected before birth. As well as housing, the contaminated environment can include pastures that have been grazed by cattle or have been spread with slurry, and also fodder made from those pastures.
Infection progresses slowly. For two years or longer, infected animals usually remain without apparent disease and are test negative. Progression of infection to clinical disease is usually triggered by stress, most commonly due to peak milk output in the second to fifth lactations but other stressors such as calving, movement to a new herd, bullying, and concurrent disease may be factors.
Faecal shedding of MAP increases as infection progresses, but can occur before disease becomes apparent. Within a herd, spread of infection is typically slow at first, but gradually increases as the infection pressure increases and more calves become infected each year. Key actions to controlling infection are identifying high-risk animals (mostly test-positive animals) for isolation and removal, and hygienic conditions for calving and calf-rearing. The key to preventing infection is to maintain a fully closed herd. For more information, visit: https://animalhealthireland.ie/assets/uploads/2021/07/JD-Fact-Sheet-2021-10-Reasons-to-join-IJCP-FINAL.pdf?dl=1
National BTM testing
The Department of Agriculture, Food and the Marine (DAFM) has been conducting a national screening programme each spring and autumn from 2019 on bulk tank milk (BTM) samples for a range of animal diseases, including JD. Although relatively insensitive, the detection of antibodies is a cost-effective method for monitoring and controlling diseases such as JD.
All dairy processors provide milk samples from each milk supplier to DAFM. Two rounds of testing are performed each year, during spring and autumn with analysed results currently available up to autumn 2022 (eight rounds of testing) (Figure 1), although samples have not been available for testing from all herds for all sampling rounds.
Results so far in the period 2019 to 2022 show that for each round of testing two to seven per cent of herds are deemed to have a positive BTM sample (occurrence of antibodies against MAP at a detectible level). This is considerably lower than the estimated overall herd infection prevalence in the national population (30 per cent). This disparity arises due to the fact that the proportion of cows with antibodies in positive herds falls below the threshold necessary to consistently yield a positive result in bulk tank testing.
As a result, a negative outcome offers little assurance that a herd is infection-free. However, a positive result is useful for case detection, suggesting that the level of infection present is at the upper end of the national profile. Peer-reviewed studies indicate that the sensitivity of the BTM test for MAP is on average low at 16 per cent3 but that increasing levels of herd prevalence (>nine per cent herd prevalence) are associated with better performance of the test in the form of increased test sensitivity3. In terms of specificity (ability of the test to categorise uninfected herds as negative), figures quoted for the test are 95 per cent.
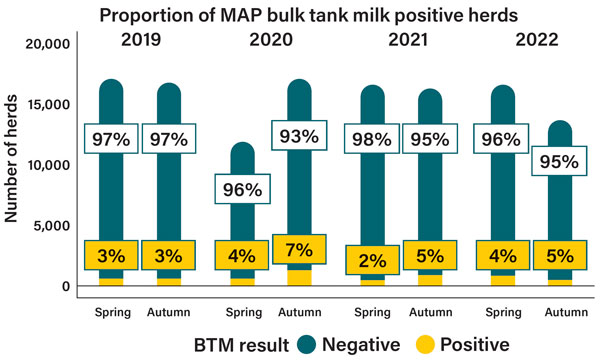
Figure 1: Proportion of BTM samples tested, and associated results, in national surveillance from 2019 to 2022 inclusive.
This means, overall, the BTM test will pick out 16 per cent of infected herds, but alongside this also disclose five per cent of MAP-uninfected herds as false positive results. It is however important to note that, rarely, factors such as recent TB testing or exposure to closely-related bacteria in soil or water may cause false positive results. Therefore, a positive BTM result should be interpreted carefully, in consultation with the herd’s veterinary practitioner who is familiar with the herd’s health profile.
Overall, during the period 2019 to 2022 a total of 17,182 herds have been BTM tested across any of the eight sampling points with 3,117 herds tested positive at least once. 2,256 herds have returned only one positive result between 2019 and 2022, while 861 herds have tested positive on two or more occasions (Figure 2).
Any herd which is not a member of the Irish Johne’s Control Programme (IJCP), and which discloses a positive BTM result is informed of this result in the form of a letter from DAFM. These letters were first issued in autumn 2022, with the initial letters containing the results of five rounds of testing. Since autumn 2022, letters have been issued after each spring and summer sampling round and contain all dates on which a positive BTM sample was disclosed on the farm.
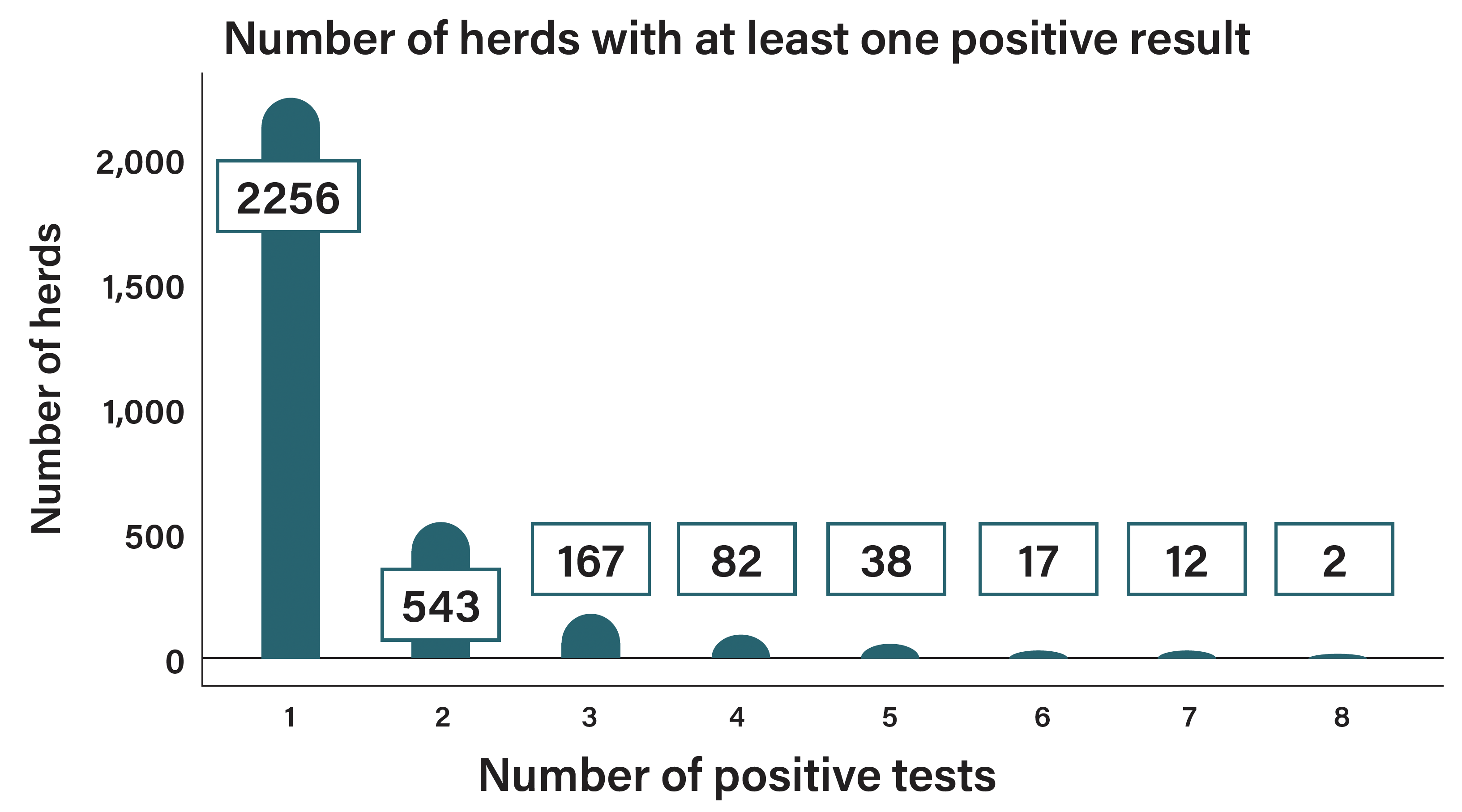
Figure 2: BTM herds and the number of times they have tested positive in the eight sampling rounds between 2019 and 2022.
BTM positive herds in the IJCP
Each herd which receives a letter from DAFM informing it of a positive BTM result also receives an information leaflet from Animal Health Ireland (AHI) advising the herd owner to discuss the results with their veterinary practitioner and register for the IJCP. Since September 2022, when letters were first sent to herd owners informing them that they had disclosed BTM positive result(s), 203 recipient herds have joined the IJCP (which has a current total membership of 2,231 herds).
When a herd joins the IJCP it becomes part of an integrated programme which is designed to provide control strategies for the herd owner, determine prevalence of infection and confirm presence, if faecal ancillary testing discloses as positive. The programme comprises the following components:
An annual veterinary risk assessment and management plan (VRAMP) conducted by their approved veterinary practitioner (a PVP who has completed AHI-delivered training on the programme). A VRAMP will identify risks that apply to your herd for introduction and spread of infection, and establish agreed management changes.
Annual whole herd testing (WHT) of all eligible animals (those aged two years and above) using individual blood or milk samples.
Follow-up (ancillary) testing of dung samples from animals disclosing at the ELISA blood or milk tests.
Herds that have one or more positive ancillary test results are confirmed as infected and are provided with further AVP input and advice through a fully-funded TASAH (targeted advisory service on animal health) assessment.
Current results from IJCP data show that in over 100 of the 203 herds which have joined the IJCP (post receiving a BTM letter indicating a positive result) and then completed both their ELISA herd test and ancillary (faecal) testing of individual highlighted animals confirmed JD at a rate of just under 30 per cent.
Indeed, when we look at the herd apparent prevalence (HAP) in Test Negative Pathway (TNP) IJCP herds and compare their HAP levels to those of herds which have joined the IJCP as BTM positive herds we find that the BTM positive herds are associated with a statistically significant higher HAP (Figure 3).
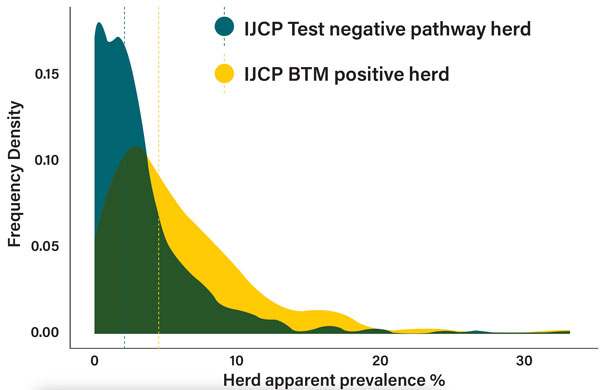
Figure 3: Distribution of herd apparent prevalence (HAP) levels in BTM positive vs Test Negative Pathway IJCP herds with median for each group included as dotted line.
Summary of BTM testing
BTM testing for JD has a low herd sensitivity (16 per cent) which means that it is unsuitable for providing assurance of freedom from MAP infection.3 One significant advantage of BTM testing is that sensitivity of the assay is greater in higher prevalence herds making it a low-cost option for detection of these higher prevalence herds.4
BTM testing functions as a low-cost screening test meaning that positive results must be followed up with a whole herd test (WHT) before any conclusions can be drawn. This follow-up testing involves individually ELISA-testing (milk or blood) all animals in the herd over two years of age and applying confirmatory ancillary (faecal PCR) testing to get a full epidemiological picture of the herd. Given knowledge of the epidemiological picture of the herd, application of the VRAMP/TASAH can provide the herd owner with the effective practices tailored to their herd to help mitigate JD on their farm.
Additional information and how to register for the IJCP
Additional information about BTM testing and how to register for the IJCP are available on the AHI web site which can be accessed at the following link: https://animalhealthireland.ie/programmes/johnes-disease/bulk-tank-milk-testing-for-johnes-disease/
You can also contact Animal Health Ireland for information or to register for the programme, your veterinary practitioner for herd-specific advice, or your co-op’s milk quality advisor.
- Lanigan GJ, Hanrahan K, Richards KG. MACC 2023 : An Updated Analysis of the Greenhouse Gas Abatement Potential of the Irish Agriculture and Land-Use Sectors between 2021 and 2030 Prepared by Teagasc Climate Centre. 2023;(July).
- Skuce Philip. Acting on methane: Opportunities for the UK cattle and sheep sectors [Internet]. 2022. Available from: www.ruminanthw.org.uk
- Delafosse A, Meens E, Rambaud T, Hanoy F, Achour H. Sensitivities of a bulk-tank milk ELISA and composite fecal qPCR to detect various seroprevalence levels of paratuberculosis in cattle herds in Normandy, France. Can Vet J [Internet]. 2019 Mar [cited 2019 Mar 22]; 60(3):275–81. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30872850
- Sergeant ESG, McAloon CG, Tratalos JA, Citer LR, Graham DA, More SJ. Evaluation of national surveillance methods for detection of Irish dairy herds infected with Mycobacterium avium ssp. paratuberculosis. J Dairy Sci. 2019 Mar 1;102(3):2525–38.
1. Given application of the JD BTM test on one occasion (sensitivity=15 per cent/specificity=95 per cent) to 20,000 herds nationally where JD herd prevalence is at 30 per cent (6,000 herds), how many true positive herds would you expect the test to pick out?
A 1,000
B 900
C 800
D 700
2. Given application of the JD BTM test on one occasion (sensitivity=15 per cent/specificity=95 per cent) to 20,000 herds nationally where JD herd prevalence is at 30 per cent (6,000 herds), how many false positive herds would you expect the test to pick out?
A 1,000
B 900
C 800
D 700
3. Based on the results of your JD BTM testing of the 20,000 herds nationally in questions 1 and 2, what would you estimate the overall national herd prevalence to be?
A 10 per cent
B 8 per cent
C 6 per cent
D 4 per cent
4. What is the effect of a higher herd prevalence on the sensitivity of the JD BTM ELISA test?
A It reduces the sensitivity of the BTM ELISA test significantly
B It reduces the sensitivity of the BTM ELISA test marginally
C It does not affect the sensitivity of the BTM ELISA test
D It increases the sensitivity of the BTM ELISA test
5. Which is the true statement about the JD BTM test as applied in Irish dairy farms?
A With a negative BTM test, the interpretation is that the herd is definitely free from JD
B With a positive BTM test, the interpretation is that the herd is definitely JD infected
C The BTM test is a useful method for epidemiologists to determine the prevalence of JD in the national dairy herd
D The BTM test is a useful method to pick out herds that are likely to be heavily infected with JD but is also likely to return a negative test result for many infected herds
ANSWERS: 1B; 2D; 3B; 4D; 5D.










