Large animal - November 2019
What can dairy farmers do to help reduce the risk of antibiotic resistance?
In this article, Finola McCoy, senior programme manager CellCheck, with the support of the Cellcheck Technical Working Group, explores some key questions around antibiotic resistance and why it is cause for concern
What role does antibiotic use on the farm play in the development of antibiotic resistance (AMR), and how can those of us working in the dairy sector positively influence this? When drying off cows, has a selective dry-cow strategy a role to play in reducing the use of antibiotics on farm? What science is available to help choose which herds are suitable for selective dry-cow strategies, and how do we best identify the cows in these herds that may not need antibiotics at drying off, while still protecting the udder health of the herd? This article draws on national and international research to answer some of these questions, while also highlighting that there are still some questions to which we don’t have all the answers.
Recap – what is antibiotic resistance?
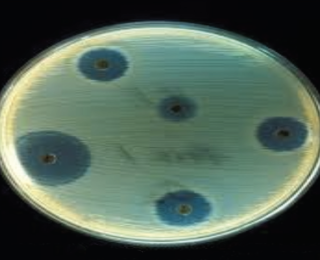
Antibiotic resistance, also antimicrobial resistance (AMR), arises when the bacteria, which cause infection, survive exposure to an antibiotic that would normally kill them or stop their growth. AMR allows strains of bacteria that are capable of surviving exposure to a particular antibiotic, to grow and spread. Resistance to one or more antibiotics may occur and this is a real threat to our planet especially when we only have a limited number of antibiotic groups to treat infections in humans and animals. We can grow bacteria on an ‘agar plate’ and use different antibiotic discs to check which antibiotic works best. Where there is a zone of ‘no growth’ around the disc, we know the antibiotic works. Where there is bacterial growth around the disc, then the bacteria are resistant to that antibiotic. See Figure 1.
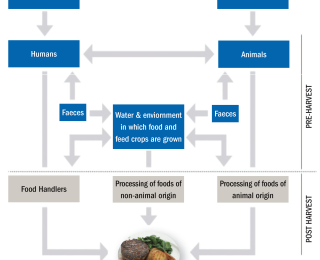
Not only do we see bacterial infections on farms that are difficult to treat because of resistance, but we also encounter human infections resistant to antibiotic treatment. Worse still, bacteria that are resistant may spread and share these traits with other bacteria including those in the environment, eventually tracking back to the bacteria that cause infections in humans. The pace at which AMR has been developing in more recent years has increased, and it is now recognised as being a significant threat to human health. See Figure 2.
The importance of antibiotics in human and animal health
AMR is responsible for an estimated 25,000 deaths and €1.5 billion in extra healthcare costs every year in the EU alone. Hence, the sense of urgency about addressing this issue and doing so at a global level. AMR is linked to antibiotic use – increased antibiotic use in both humans and animals is associated with an increase in AMR. In relation to mastitis-causing pathogens, there is evidence to show that different bacterial species develop resistance to different antibiotic groups at different rates.
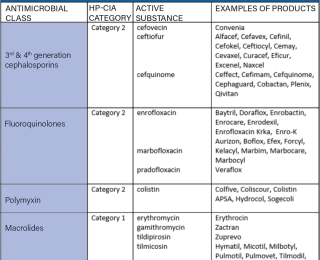
Currently, antibiotics are used by doctors to treat sick people and used in the agricultural sector to treat animals. In recent years, there has been increasing recognition of the linkage between AMR in people and antibiotic use in animals. For these reasons, there is increasing scrutiny of the use of antibiotics in the agricultural sector. There is agreement on the importance of antibiotics to treat sick animals. However, it is no longer considered acceptable that antibiotics should be used to prevent disease, particularly when there are other proven strategies. The type of antibiotic used for treatment of animals is also an important consideration. ‘Highest Priority Critically Important Antibiotics’ (HP-CIAs) need to be preserved for human use. The Department of Agriculture, Food and the Marine (DAFM) recently published guidelines on the use of CIAs in animals (Table 1). Disease prevention and improved herd health certainly help reduce the need for antibiotic on farm. Vets and farmers need to be mindful to avoid using those antibiotics that are classified as HP-CIAs.
What can CellCheck and the dairy farmer do to reduce AMR?
The focus of the CellCheck programme is on improving herd and udder health, thereby minimising clinical and subclinical disease. This also reduces the need for mastitis treatments, both injectable and intramammary.
The role of antibiotic dry cow therapy
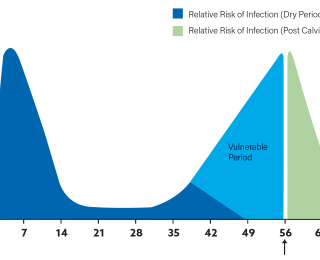
The practice of dry cow therapy is being questioned in many countries by farmers, consumers and society in general. Antibiotic dry cow therapy undoubtedly has an important role to play in treating infections that persist at the end of lactation and maximising cure rates. It has also traditionally been used to prevent new infections occurring over the dry period. While the dry period is a high-risk period for udder infections both clinical and subclinical, the consumer at home and abroad is becoming intolerant of using antibiotics on a ‘just-in-case’ basis. Considering our changing attitude and approach towards the use of antibiotics in a ‘preventative’ fashion, do we also need to rethink how and why we use dry cow therapy? And in fact, how do we define dry cow ‘therapy’?
Recent analysis of sales data in Ireland (More et al., 2017) indicated that sales of dry cow intramammary antibiotics were sufficient to treat 100% of the national milking herd, ie. all quarters of all cows are being treated at the end of lactation. This is what is referred to as ‘blanket dry cow therapy’, which until recently was recognised as best practice in mastitis control and has made a very positive contribution to udder health in many countries. However, as we learn more about AMR and what drives it, we need to review what is considered best practice, as well as the implications of modifying those ‘traditional’ recommendations. Change is not without risk.
What are the risks and benefits of moving away from blanket dry cow therapy?
An alternative to blanket dry cow therapy is a ‘selective’ dry cow strategy. A selective dry cow strategy involves administering internal teat sealant only to a selected proportion of suitable cows at drying off, with the remainder of the cows receiving both an antibiotic tube and an internal treat sealant. While this is considered a more prudent use of antibiotics and would reduce antibiotic use on many farms, we need to bear in mind that this practice is not without risk. So how can we manage this risk? The CellCheck Technical Working Group recently reviewed all of the science and research on dry cow therapy published since the early 2000s, and have identified the following key risks:
1.
- The first risk is of introducing bacteria when we infuse any intramammary tube into a quarter. When we use internal teat seal only, there is no antibiotic present as ‘backup’ and so the potential consequences are even greater. These ‘introduced’ bacteria can cause severe cases of mastitis, sometimes resulting in death, early in the dry period. In addition, many of the cases of mastitis in early lactation have been shown to have a dry period origin. Hygiene standards and practices at drying off – as outlined in detail in the CellCheck Farm Guidelines (pages 117-119) – are essential to protect the udder health of the uninfected cow. Hygiene and management of the dry cow environment is also crucial
- The second risk is of missing the opportunity to cure quarters that were infected at the point of drying off in order to maximise cure rates before the next lactation starts. A very common question is ‘how do I know which ones are the infected animals?’ There are many criteria that need to be considered when making these decisions, including milk-recording results and milk-culture results. Even with all this information on hand, further questions remain such as ‘how many milk-recording results do I need to have and how close to drying off do they need to be?’