Focus - June 2020
Alternatives to antibiotics? Eubiotics, the microbiome and immunity
With the ever decreasing list of currently available antibiotics, the growth of resistance to them, and the dearth of new and novel antibiotics arriving into the marketplace, it has never been so timely to develop and deliver new, effective and proven alternatives to traditional antibiotic substances. In a comprehensive report, Dr Tom Barragry PhD MSC MVB MVM MRCVS Dip ECVPT, introduces us to eubiotics as one such alternative
Click on image to view
AMR – problems and consequences
Antimicrobial resistance (AMR) is now a major global concern and has been identified as one of the most serious threats to the future maintenance of human and animal health.
The World Organisation for Animal Health (OIE) ,World Health Organization (WHO) and the EU Commission have become very vocal on this issue and have published numerous position papers and recommendations on this topic, with a view to avoiding and minimising this growing threat of AMR. The EU in its One Health Initiative and Horizon 2020 programmes has focused on the need to restrict and limit the therapeutic usage and the indiscriminate usage of antimicrobials in animals and they have emphasised the urgent need to develop alternatives to antibiotics in veterinary medicine and in animal husbandry. The EU has encouraged Member States to assist in the funding of companies and research institutions in finding ways to circumvent the use of antibiotics, through the research and development of viable non-antibiotic alternatives.
The new Veterinary Medicines Regulation (2019/6 of December 11, 2018) has spelled out the need to restrict antimicrobial usage on farms and has banned the metaphylactic and prophylactic use of antimicrobials in animals especially by the oral route. This Regulation will come into effect on January 28, 2022.
Some of the aims of the new Veterinary Medicines Regulations include, inter alia:
- Control of AMR in the EU as it relates to the use of veterinary antibiotics;
- Ban of the prophylactic/metaphylactic use of antibiotics in farm animals by 2022; and
- Encourage funding of industry to develop safe and proven alternatives to antibiotics.
At political and consumer level, there is a global demand and desire for:
- Green, safe and sustainable food production; and
- Alternatives to assist global health and reduce AMR.
Because of the serious implications of antibiotic resistance for human health via cross resistance and transferable drug resistance from pathogens of animals to human, or via commensals in the gut, the OIE and WHO, in 2018, drew up its sixth revised list of critically important antibiotics (CIAs) in human medicine. In that document, they set out a programme for the avoidance of the use of these critical antibiotics in animal therapy, insofar as it is possible. It also provided a ranking system categorising the stringency and limitations of usage of such CIA antimicrobials in veterinary medicine.
In short, with the ever decreasing list of currently available antibiotics, the growth of resistance to them, and the dearth of new and novel antibiotics arriving into the marketplace, it has never been so timely to develop and deliver new, effective and proven alternatives to traditional antibiotic substances.
Eubiotics – alternatives to antibiotics in the future?
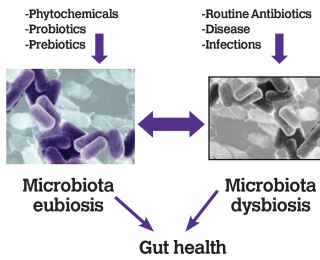
As the routine use of antibiotics in livestock is no longer acceptable, and prophylactic/metaphylactic use will be phased out over the coming years, the term ‘eubiosis’ has made its entry. It is a completely new way of approaching animal health: from fighting the microflora with antibiotics, towards supporting and cooperating with the favourable microflora. Eubiosis refers to an optimal balance of the microflora of the gastrointestinal tract. It is one of the key factors for optimal animal performance and development of immunity. Feed additives proven in this regard can be a very helpful tool to promote eubiosis and to overcome an imbalance in the microflora of the gastrointestinal tract, also known as ‘dysbiosis’.
Notwithstanding all this, there is no real change as regards one key role of antibiotic usage. Antibiotics, undoubtedly, will always remain to play a key role as potent therapeutic agents to be very judiciously used in severe clinical outbreaks of infectious disease. In terms of drug potency, as yet there are few viable pharmacological alternatives to replace this fire brigade therapeutic approach.
What has changed however, is the strategy towards reducing overall antibiotic usage via better prophylactic measures leading to fewer outbreaks of clinical disease and, hence, a reduced need for antibiotic intervention.
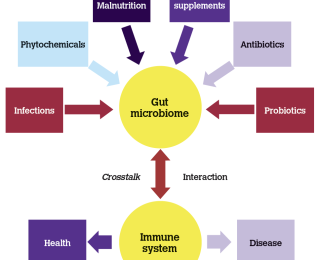
This approach will involve better biosecurity measures better prophylaxis, including vaccines, and the regular deployment of better eubiotic agents, such as proven probiotics prebiotics and proven phytochemicals to promote a healthy gut and the promotion of better immunity. All of the above are preventive agents given from birth as a pre-emptive strike to head off disease.
Gut microbiome
Probably no such other scientific area has received more collective attention and focus in recent years, resulting in a plethora of international scientific publications (particularly in the human medical zone), than the science of the gut microbiome. Research has revealed its surprisingly potent effect on general immunity and the necessity of maintaining a good eubiosis in order to ward off disease in all body compartments (not just the gut). This research area and its scientific revelations have now spilled over into the veterinary medicine domain with a consequent and commensurate increase in the practical applications of this science for phytochemicals and probiotics acting on the gut.
This spike in gut research in the animal feedstuffs sector has been triggered especially by the growing problems of AMR, the push back on antibiotic usage in the EU and the urgent imperative to find viable and proven effective alternatives to antibiotics in young animals. The global eubiotics market is projected to reach a value of USD $7.47 billion by 2022. The demand for eubiotics in animal feed is projected to grow very significantly during the forecast period, owing to the rapidly increasing global demand for quality animal products free from antimicrobials.
The eubiotics market is influenced by the growth of the awareness among consumers with respect to preventive healthcare and nutrition. The biotechnology industry is now creating an opportunity for growth in the eubiotics sector with a variety of good preparations.
Derived from the Greek words ‘eu’ meaning healthy and ‘bios’ meaning life, eubiotics is the science of healthy living via a healthy gut. The term is now used in the animal feed industry where promoting growth and maintaining gut health and immunity at the calf hood stage are the main requirements to ensure future productivity as adult animals. Eubiotics refer to a healthy balance of the micro-flora in the gastrointestinal tract. The primary reason for using eubiotics is to sustain the intestinal eubiosis, which will result in an improved health status and performance in farm animals. Also, the availability of the required balance of eubiosis optimises nutrient absorption.
Eubiotics include feed additives such as polyphenols, plant-derived natural substances (phytochemicals) combined with organic acids, probiotics, prebiotics and essential oils.
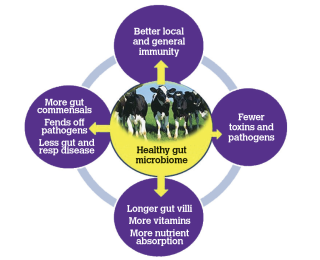
It has been long known, of course, that a healthy gut and a balanced gut microflora prevent gastrointestinal tract (GIT) problems such as diarrhoea and vomiting and can optimise nutrient absorption and food utilisation. Probiotics have been well established and used in this fashion in animals and man, for over 20 years now, to promote a balanced gut microflora and maintain gut health. However, what is not generally known nor appreciated by many people, and what has emerged from very extensive research in human medicine in latter years, is that the gut microbiome exerts a very potent effect on the general immune system also and that there is a high correlation between a dysbiotic (malfunctioning) gut microbiome and disease occurrence in other body systems such as the respiratory tract and the central nervous system (CNS), because of sub optimal systemic immunity. Hence, a healthy gut microbiome in young animals not only maintains gut health but also can have a protectant effect against the occurrence of other diseases (ie. outside of the GIT effect). Indeed, a very cursory look through the literature journals and scientific publications will show that the number of international conferences and seminars on the potential immunological role of the gut microbiome has grown exponentially over the last three years. The gut-lung axis has now been extensively referenced in the human literature, and many papers now show a connection between the gut microbiome and infections in the respiratory tract, auto immunity and brain tissue.
It is interesting to note that in the veterinary domain, proven plant poly phenols and proven probiotics not only benefit calf health and keep neonatal scours to a minimum, but also reduce the incidence of respiratory infections in calves.
Microbiome crosstalk
New science is only beginning to unravel the extent of the mutual influence and interaction (the ‘crosstalk’) between the gut microbiome, the respiratory tract and immunity. It is already clear that this is very significant and has a huge impact on health and on the body’s homeostasis – the state of balance, of dynamic equilibrium that is fundamental for survival. Human medical research is now replete with many evidence-based and substantiated scientific reports in this regard. The gut microbiota influence many aspects of physiology, from metabolism, to the cardiovascular system, the respiratory system, and the nervous system. Dysbiosis of the gut microbiome is caused by the imbalance between the commensal and pathogenic microbiomes. The commensal microbiome regulates the maturation of the mucosal immune system, while the pathogenic microbiome causes immunity dysfunction, resulting in disease development. Thus, commensals have a huge role to play, not only in vitamin synthesis but in immunity development also.
The immune system is the group of cells and molecules that protect from disease by monitoring the body and responding to any foreign (non-self) substances they perceive as threats, particularly infectious microbes. The immune system has co-evolved along with a diverse gut flora, not only to create defences against pathogens, but also to develop tolerance for beneficial microbes. The dialogue between the immune system and the microbiota begins at the moment the body gets in contact with microbes at birth. During growth and development, the microbiota shape the development of the immune system, and vice versa, the immune system shapes the composition of the microbiota. This communication and mutual regulation is maintained throughout life and is the key for a healthy interaction between the microbiota and the immune system.
Immunity of the gut microbiome system
All mammals harbour complex and dynamic populations of microorganisms (known as the microbiota), which are made up of bacteria, archea, fungi, viruses, and protozoa and colonise mucosal surfaces, such as those found in the gut, nose, mouth, airways, and urogenital tract, as well as non-mucosal surfaces, like the skin. However, the intestine holds the largest collection of microbes, with a total of ~1014 bacterial cells. In fact, in man, the entire population of bacterial genomes in the microbiota contains >5 million genes, outnumbering the number of human genes by orders of magnitude. The gut is home for a very large number of microbes and in a human, they number around 38 trillion microbes, at least as many cells as there are in the entire human body. Their collective genome, known as the gut microbiome, contains 150 times more genes than the human genome. The body’s immune system has co-evolved along with a diverse gut flora, not only to create defences against pathogens, but also to develop tolerance for beneficial microbes. As a consequence, the immune system and the gut microbiota developed a mutualistic relationship, regulating one another and cooperating to support each other. The importance of this interaction is clearly highlighted by the fact that 70-80% of the body’s immune cell drivers are found in the gut. While it is clearly undeniable that antibiotic therapy is an invaluable clinical tool, an increasing number of studies have demonstrated that perturbations to the gut microbiota by oral broad-spectrum antibiotic treatment results in alterations to the functions of the microbiota in ways that are, ultimately, detrimental to host health via an impaired immune system. For example, disrupting the gut microbiota with unnecessary broad-spectrum antibiotics during neonatal periods may alter the development of the immune system. Furthermore, some studies in human medicine now suggest that regularly treating individuals with antibiotics, particularly broad-spectrum antibiotics, has some negative consequences on the immune system, and can lead to increased susceptibility to infections in different body compartments. This is a wholly separate issue from the development of a significant AMR problem also.
In a recent paper published in September 2019 in Cell, researchers at Stanford University found that oral antibiotics that can kill gut microorganisms, can alter the human immune response to seasonal influenza vaccination. The researchers also found that people who took antibiotics experienced changes to their immune systems that promoted a pro-inflammatory state. Thus, alternatives to antibiotics appear to be better for encouraging the development of the immune system via the naturally balanced bacterial population of the gut microbiome.
Commensals have long been known as being beneficial to the gut by way of vitamin and protein synthesis and production of beneficial growth factors. However, it now seems that commensals have another important role the governing of immune function.
Commensal microorganisms are required for the maturation of the immune system, which ‘learns’ to differentiate between commensal bacteria (which are becoming almost quasi-self and tolerated antigens) and pathogenic bacteria. Toll-like receptors (TLRs) from the membrane of the epithelial and lymphoid cells of the small intestine are involved in this differential recognition, being responsible for the normal development of the intestinal mucosal immune system. TLRs suppress the occurrence of an inflammatory response and promote immunological tolerance to normal microbiota components. The role of TLRs is to recognise different general microbe-associated molecular patterns (MAMPs) containing various bacterial antigens (eg. peptidoglycan components-muramic acid, capsular polysaccharides and lipopolysaccharides, flagellin and unmethylated bacterial DNA CpG motifs) and to trigger the innate intestinal immunity. Research in human medicine has now established links between the immunity governed by the gut microbiome and the occurrence of influenza, respiratory disease, asthma, COPD, atopy and possibly Parkinson’s disease, along with many other deleterious conditions and infections.
Tight junctions and the intestinal wall
The intestinal wall is the primary interface between the gut microbiota and the body. It acts as a dynamic barrier that isolates the body from gut microbes but allows desirable interactions to take place. The intestinal barrier is made up of physical and chemical elements. The physical barrier is created by the epithelial cells that line the gut, the molecules on their surface, and the mucus they produce; the chemical barrier is created by inflammatory molecules (cytokines), antibodies, and antimicrobial substances produced by epithelial and immune cells.
Epithelial cells recognise microbial products via immune receptors known as pattern-recognition receptors (PRRs). Activation of PPR enables a dynamic adjustment of epithelial activity based on chemical signals from the microbiota. This allows epithelial cells to adjust their antimicrobial response to eliminate pathogenic infections, destroy infected cells, and influence the composition of the gut flora. Proper PRR signalling is important for the maintenance of tolerance to good microbes, for the elimination of intestinal infections, and consequently, for the maintenance of a balanced gut microbiota.
When there are faulty tight junctions in the GIT, many microbial pathogens, toxins and metabolites cross the epithelial barrier and are absorbed into the blood, through which they can reach other tissues in our body causing disease. On the other hand, some commensal-based microbial metabolites can influence the development, maturation, and function of circulating and tissue-resident immune cells in different organs, including the brain. Through these metabolites, the gut microbiota is able to fine-tune innate immune responses throughout the body.
The barrier function of the intestinal wall is also maintained by immune cells. For example, microfold cells (M cells) are cells of the immune system found among epithelial cells that transport microbes and microbial antigens across the epithelial layer and deliver them to antigen-presenting cells to initiate immune responses. Found within the intestinal wall are also dendritic cells, whose extensions protrude from between epithelial cells into the intestinal lumen, where they probe the microbiota environment, searching for potential threats and devising adequate responses. T and B cells located in the intestinal wall participate in adaptive immune responses that contribute to the maintenance of the epithelial barrier and to the suppression of responses to harmless microbes, thereby promoting immune homeostasis. Again, the distinction between self and non self, pathogen and commensal is crucial.
Healthy intestinal barrier function allows certain gut-derived molecules to get into the body, while keeping others out. This supports better immune and systemic performance. The nature of the feed intake influences many aspects of the microbiotica-immune system crosstalk, including, for example, the permeability of the intestinal barrier.
Eubiotics on the veterinary market
One of the biggest problems for veterinarians and stock owners to date in endeavouring to select a good eubiotic as a viable alternative to an antibiotic, has been the number of unproven, unsubstantiated and substandard products on the market. Many probiotics have been of very poor quality with no proven in vitro nor in vivo effectiveness. In addition, many of the marketed polyphenol type of plant-based phytochemicals have little or no research and development behind them, are unproven formulations and simply lean on the generic public literature to support their nonspecific claims. There are, in fact, over 8,000 polyphenols and flavonoids in nature, so it’s not simply a question of quoting the literature for scientific support when, in fact, the manufacturer may not actually know which precise polyphenol or flavonoids or organic acids are in their formulation and in which precise formulation and combination.
However, against all that, it should be said that there is very sound scientific evidence that certain eubiotics such as proven polyphenols and proven probiotics can have very beneficial effects on the gut microbiome and on general immunity status resulting in a reduction in antibiotic usage, less disease in calfhood, and better weights at weaning. A wide number of calf, poultry and pig producers have now pushed back on antibiotic usage and are reporting excellent growth and health results with in-feed polyphenols and organic acids and also with licensed/authorised probiotics. There are also a few published papers and field trials with a number of these products showing greater health and growth rates in young calves. A feature of some of these, mainly via an immune action, is the lessened incidence of cryptosporidia and Escherichia coli (E. coli) infection in young calves when fed certain polyphenols/organic acid phytochemicals, along with certain potent probiotics.
Phytochemicals such as probiotics, polyphenols/organic acids, in the main are not curative nor are they therapeutic. They are preventative agents which give best results when fed from birth onwards, in order to strengthen the gut immunity and populate a healthy gut microbiome, in advance of an infectious challenge which in many cases are surprisingly well repelled.
Anti-inflammatory effects, antioxidant properties, antimicrobial effects, and sealing of tight junctions together with enhanced intestinal villus length and better villus:crypt ratios under pin the efficacies of some of these products and this has been well documented in some peer-reviewed scientific papers.
The veterinary eubiotic market is undoubtedly a growing market sector, as a viable replacement and alternative for antibiotics. Without question, more eubiotic products and combinations thereof will appear on the European market and these could have a major beneficial and significant role in promoting animal health from birth. However, the usefulness of these products is in direct proportion to their published and proven in vitro and in vivo effectiveness.
Recent US research
Recent US research
Recent scientific literature provides evidence that high nutrients and disease-free status pre weaning in calves has a positive imprinting effect on growth and subsequent lactation performance. Imprinting of future development of mammary gland tissues occurs before weaning in calves. Antibiotic treatment pre weaning in calves has been shown to retard subsequent milk production in cows when it is compared to milk production in cows where the calves never received antibiotics pre weaning. This ‘lactocrine imprinting’ is now a well-accepted phenomenon, and is positively affected by increased growth rates, high nutrients and high immunity in calfhood up to weaning.
Recent literature provides evidence that high nutrient pre weaning feeding of calves has an imprinting effect on growth and lactation performance (Bach, 2012; Soberon, 2012). Growth rate due to calf starter intake is correlated with future milk yield and survivability to second lactation. It is suggested that for every 100g of average daily gain during the first two months of the calf’s life, an additional 225kg of first lactation milk yield could be expected (McGuirk, 2016).
It would also appear that the less antibiotic used in calves pre weaning (and the less disease that occurs) positively affects downstream performance, milk production, and milk capacity subsequently in the adult animal. Published evidence shows that high immunity in the pre weaning stage, a disease-free status and high nutrition in pre-weaned calves, increases the future mammary gland parenchymal tissue development (site of casein synthesis). A number of peer-reviewed papers have shown this:
Calves treated with antibiotics give less milk
A study showed no difference in first-lactation milk production between calves with or without diarrhoea; however, scouring calves treated with antibiotics gave 1,086lbs less milk during their first lactation than those not treated (Soberon et al, 2012). These data do not indicate that antibiotics should not be used when needed, but that prolonged damage to the intestinal tract or other complications may occur. Besides scours, respiratory disease is a common problem in calves. McGuirk (University of Wisconsin) estimates that three to four times more calves are affected by respiratory problems than are treated or identified by producers. Studies have shown decreased growth, decreased survival to first calving, and increased age at calving in calves with respiratory disease within the first 60 days following movement to group housing (Stanton et al, 2012). Vaccines, minimising stress, and providing proper ventilation in the microenvironment around the calf without causing drafts are important components in preventing respiratory illness (Amaral-Phillips, 2018).
Examples of proven eubiotics
Examples of proven eubiotics
This author is familiar with two useful eubiotics, which have been well proven in vitro and in vivo, and have been highly successful in field trials. A brief summary of these eubiotics follows.
1 Veterinary-licensed probiotic bacteria
When choosing a probiotic, veterinarians should select a product classified as 'medicine', as it will have been assessed by a regulatory agency under safety, efficacy and quality. The therapeutic effects are much higher for this type of approved product and critically, the levels are guaranteed to be above a specified level at the end of the shelf life, provided it is stored correctly. The only veterinary-licensed probiotic available in the UK and Republic of Ireland is a probiotic bacteria aimed at preventing diarrhoea in young calves. Considerable independent trial work has been carried out on the benefits associated with probiotic use in recent years and the rationale behind using probiotics is summarised below.
Competitive exclusion in the digestive tract
Providing high levels of beneficial bacteria interferes with the attachment of enteric pathogens, such as E coli, to the gut wall. The objective is to crowd out the pathogens and prevent their establishment within the gut.
Antimicrobial effects
Probiotics are known to produce lactic acid and bacteriocins which have been shown to have an inhibitory effect on some pathogens.
Lactic acid production
As the pH in the gut is reduced through the production of lactic acid, the environmental conditions within the intestine become unfavourable for some pathogens. The more acidic environment encourages increased enzymatic activity, which improves digestion.
Immune stimulation
Probiotics can stimulate the body’s immune response system directly and indirectly via the gut microbiome.
Studies
This probiotic bacterial product has been shown, in independent field trials, to help calves up to the age of 12 weeks to resist pathogenic infection, thus avoiding the enteric upsets which cause diarrhoea. Treatment with it has shown a significant reduction in the incidence of scouring in calves, resulting in improved growth rates and body condition. Observations by the author noted that it worked as effectively as an antibiotic in helping to treat calf scour. Also, when calves suffering from pneumonia were observed, they recovered more quickly, which is likely due to the immune stimulation effect.
2. Phytochemicals
A new Irish biotech company, Auranta, which recently received an Innovation of the Year Award, has focused on the development of a suite of plant-based phytochemicals with a wide range of proven physiological and protective actions. These unique compounds have been formulated as natural additives for use in calves, poultry and pigs. The Auranta product has six international peer-reviewed publications, which have documented the following: increased immunity; increase in gamma interferon IFN-y; sealing of gut tight junctions; anti-inflamatory effects; antioxidant effects; antimicrobial effects; and a positive impact on the gut microbiome. Field trials and clinical uses have corroborated these actions. They are intended to be used prophylactically as production enhancers via promotion of gut health and fed from birth as alternatives to antibiotics.
The formulations are based on a unique mix and combinations of natural polyphenols with the addition of specific organic acids and other plant-based extracts to provide a symbiotic effect from the entire range of ingredients. Polyphenol products have been widely described in the peer reviewed scientific literature in their various roles as antioxidants, immunity enhancers, microbiome stabilisers and numerous other biological actions. They do this via a natural physiological action, and they are not synthetic chemicals. In short, these natural products help the animal to help itself.
Disclosure
The author has acted as an independent adviser to Auranta Ltd, and has been involved with Provita Ltd and Auranta Ltd in the scientific evaluation and field assessment of their products.
References available on request.