Farm animal - October 2019
The benefits of early calf vaccination against BRD
Cara Sheridan MVB MRCVS Cert DHH, ruminant technical vet MSD Animal Health, explores whether we are acting in time to control bovine respiratory disease in our herds
Bovine respiratory disease (BRD) is the most common disease entity in calves aged greater than one month of age. Neonates (calves less than one month of age) are also affected; after diarrhoea and septicaemia, BRD accounts for 11% of mortality in this age group.1 The high incidence of this disease complex is far from a new phenomenon and yet there is little evidence of significant decline.2 BRD control is most effective when implemented in advance of occurrence of disease. A holistic preventative programme including a tailored vaccination strategy is more likely to yield satisfactory and comprehensive protection3. The question is – are we acting in time to control pneumonia in Irish herds?
Vaccination early in a calf’s life can play an integral part in keeping the present and future impact of BRD to a minimum. However, vaccination alone cannot carry the responsibility of disease control. Management of the environment, nutrition, husbandry practises and control of concurrent disease all contribute to immune competence of the calf while keeping stress and the challenge of infection to a realistic level. As with successful vaccination strategies in any species, population immunity not individual animal protection should be the aim. Timing of vaccination is also key; before the period of risk rather than implementation when disease is already established.
What are the benefits of early calf vaccination?
BRD can affect calf populations from a young age. As stated previously, 11% of neonates die because of pneumonia.1 Pre-weaning growth rates were found to decrease by eight percent for calves experiencing pneumonia and by up to 29% for calves affected by both diarrhoea and pneumonia.4 To capture the feed conversion efficiency of early life, disease must be managed and this in turn will have long-term benefits on productivity.5
Herd immunity
Herd immunity or population immunity is a form of indirect protection from infectious disease that occurs when a large percentage of a population or herd has become immune to an infection, thereby providing a measure of protection for individuals who are not immune.6,7 To put this definition into context for calf populations, early vaccination of calves will help protect those individual calves who are not yet old enough for vaccination or immunocompromised for another reason (inadequate passive transfer1 or presence of concurrent disease). However, herd immunity is vulnerable to the free rider problem.8 Individual animals which are not vaccinated free ride off the herd immunity created by those which are immune. As the number of free riders in a population increases, outbreaks of preventable disease become more common and more severe. This is particularly evident in the context of a dairy calf population where the heifer calves are vaccinated, and the bull calves are not vaccinated.
Reducing subclinical disease
Firstly, what is the prevalence of subclinical disease? The answer is essentially unknown and varies from farm to farm. In one study the prevalence of subclinical disease was found to range from 23-67%. Thoracic ultrasound was used and found to be a quick, accurate and practical approach for the diagnosis of subclinical BRD.9 Interestingly, lung consolidation (defined as a lesion greater than or equal to three centimetres in diameter) was detected by ultrasound as early as 12 days of age in calves.10 The prevalence of subclinical disease is undoubtedly a precursor for an outbreak of clinical disease.11
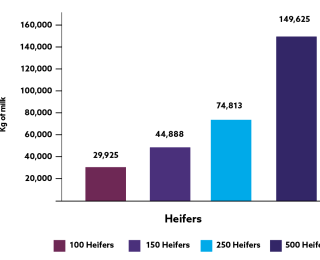
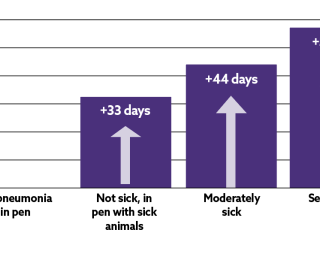
The impact of subclinical disease is well documented. From a dairy heifer perspective, the presence of lung consolidation in the first eight weeks of life resulted in a decrease of 525 kg in milk production in a 305 day first lactation10. In terms of meat, beef calves who have encountered BRD have a decreased growth performance (-61 to 108 grams per day)12 and a longer finishing period (+33 days)13, when compared to calves without a BRD history.
Reduction in antimicrobial usage
Future legislation is likely to limit prophylactic use of macrolides and metaphylactic use of both macrolides and fluoroquinolones. Furthermore, the use of oral tetracyclines is likely to be disallowed. To reduce the over-reliance on antimicrobials, strategies to reduce the incidence of disease must be sought. A study performed on multiple Dutch veal farms evaluated the influence of vaccination on antimicrobial use and performance. Vaccination significantly reduced the daily treatments with antibiotics by 14.5% and the highest level of reduction was seen in the first two and a half months on the farms14. Vaccination offers both a cost-effective solution to diseases such as BRD and supports a more conscientious use of antibiotics15.
Acceptable welfare standards
As one of the five freedoms; good health (free from pain, injury and disease) is necessary for an acceptable standard of welfare. It is also fundamental for social acceptability. Health and welfare are the two most significant issues affecting consumer trust and perception of animal-based diets according to the Eurobarometer study in 2015 (94% of surveyed consumers believe it is important to protect the welfare of farmed animals).
The advisory role of the veterinary practitioner
The practising vet has a pivotal role in implementation of a BRD control programme. As a sector we must benefit from the feed efficiency of early life, we need to focus our attention to reduce the disease challenge to young calves and to ensure they receive adequate immunity via colostrum feeding and through vaccination. This will allow us to improve productivity, decrease antimicrobial usage, decrease mortality and improve animal welfare.
-
All island disease surveillance report 2018. Accessed 20 Sept 2019
-
Hilton, W.M. (2014) BRD in 2014 - Where have we been, where are we now, and where do we want to go? Proceedings of BRDS Symposium - New Approaches to BRD Prevention, Management and Diagnosis. Denver USA, July 30-31st 2014, p7-11
-
Penny, C.D. (2015) The control of bovine respiratory disease (BRD)- are we making progress? VOLUME 23 PART 2, Journal of the British Cattle Veterinary Association
-
Magnier, S. 2014. The impact of early calf hood disease. Veterinary Ireland Journal 4(5):267-269
-
Bach, A. 2011. Associations between several aspects of heifer development and dairy cow survivability to second lactation. Journal Dairy Science 94: 1052-1057
-
Fine, P., Eames, K., Heymann, D.L. (2011) Herd immunity: A rough guide. Clinical infectious diseases, volume 52 (7): 911-916
-
Gordis, L. (2013) Epidemiology. Elsevier Health Sciences: 26-27
- Barrett, Scott (2014). Global public goods and international development. In J. Warren Evans, Robin Davies (eds.) Too global to fail: The world bank at the intersection of national and global public policy in 2025. World bank publications: 13-18
- Ollivett, T.L. and Buczinski, S. (2016) On-farm use of ultrasonography for bovine respiratory disease. Vet clinics of North America Food Animal Practice 32: 19-35
- Dunn, T.R., Ollivett, T.L., Renaud, D.L., Leslie, K.E., LeBlanc, S.J., Duffield, T.F. and Kelton, D.F. (2017) The effect of lung consolidation, as determined by ultrasonography, on first lactation milk production in Holstein dairy calves. Journal of Dairy Science 101: 5404-5410
- Griffin, D. (2014). The monster we don’t see: Subclinical BRD in beef cattle. Animal Health Research Reviews, 15(2), 138-141
- Delabouglise, A., James, A., Valarcher, J-F., Hagglünd, S., Raboisson, D., Rushton, J. (2017), Linking disease epidemiology and livestock productivity: The case of bovine respiratory disease in France. PLoS ONE 12(12)
- Bareille et al. 2006. Incidence et impact economique des troubles de sante des jeunes bovins lors de L’engraissement en pays de la loire. Journees Bovines Nantaises B, 30-35.
- Vahl, H.A., Bekman, H., van Riel. J., Report of the Veal Calf Vaccination Study with Bovilis Bovipast (MSD Animal Health), January 2014.
- Berge, A.C., Vertenten, G., Time to Vaccinate, Version 3, January 2018.